Abstract
Background
Galactin-3 is a cell adhesion molecule involved in tumor progression. Our aim was to examine Gal-3 expression in tumor, benign tissue adjacent to the tumor (adjacent-benign) and benign prostate specimens and correlated it with biochemical recurrence.
Materials and methods
Tissue microarrays were prepared from 83 tumor, 78 adjacent-benign and 75 benign tissues obtained from 83 patients undergoing prostatectomy for clinically localized prostate cancer. Tissues were stained using a Gal-3 antibody and immunohistochemistry. The staining was graded between 0 and 300 depending upon staining intensity and the area of staining. In 37 patients on whom there was follow-up (Mean: 57.8 months; Median: 68 months), staining intensity was correlated with biochemical recurrence.
Results
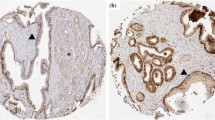
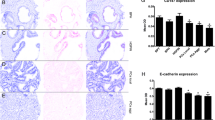
Gal-3 showed both nuclear and cytoplasmic localization in benign, adjacent-benign and tumor tissues. Median Gal-3 staining scores significantly decreased from benign (192.5) to adjacent-benign (148.8 p = 0.007) and to tumor (108.8; p < 0.0001) tissues. In univariate analysis, age (p = 0.028), Gleason sum (p = 0.007), T stage (p = 0.011), seminal vesicle invasion (p = 0.009), pre-operative prostate-specific antigen (p = 0.045) and Gal-3 staining in tumor tissues (0.018) significantly correlated with biochemical recurrence. In multivariate analysis, Gal-3 expression in tumor (p = 0.04), adjacent-benign (p = 0.037) and benign (p = 0.005) tissues significantly correlated with biochemical recurrence. Gal-3 staining in tumor tissues had 91.7 % sensitivity, 64 % specificity and 73 % accuracy in predicting biochemical recurrence.
Conclusions
This is the first study that showed a decreasing gradient of Gal-3 expression in benign, adjacent-benign and tumor tissues. Gal-3 expression may be useful in predicting biochemical recurrence.




Similar content being viewed by others
Abbreviations
- Gal-3:
-
Galactin-3
- IHC:
-
Immunohistochemistry
References
Jemal A, Siegel R, Xu J et al (2010) Cancer statistics. CA Cancer J Clin 60(5):277–300
Heidenreich A, Bellmunt J, Bolla M et al (2011) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and treatment of clinically localised disease. Eur Urol 59(1):61–71
Merseburger AS, Belka C, Behmenburg K et al (2008) Secondary hormonal manipulation. Front Radiat Ther Oncol 41:93–102
Moul JW, Merseburger AS, Srivastava S (2002) Molecular markers in prostate cancer: the role in preoperative staging. Clin Prostate Cancer 1(1):42–50
Kramer MW, Kuczyk MA, Hennenlotter J et al (2008) Decreased expression of galectin-3 predicts tumour recurrence in pTa bladder cancer. Oncol Rep 20(6):1403–1408
Merseburger AS, Kramer MW, Hennenlotter J et al (2008) Loss of galectin-3 expression correlates with clear cell renal carcinoma progression and reduced survival. World J Urol 26(6):637–642
Merseburger AS, Kramer MW, Hennenlotter J et al (2008) Involvement of decreased Galectin-3 expression in the pathogenesis and progression of prostate cancer. Prostate 68(1):72–77
Waalkes S, Merseburger AS, Simon A et al (2010) Galectin expression in urological cancer. Diagnostic, prognostic and therapeutic potential. Urologe A 49(3):387–391
Canesin G, Gonzalez-Peramato P, Palou J et al (2010) Galectin-3 expression is associated with bladder cancer progression and clinical outcome. Tumour Biol 31(4):277–285
Sakaki M, Fukumori T, Fukawa T et al (2010) Clinical significance of galectin-3 in clear cell renal cell carcinoma. J Med Invest 57(1–2):152–157
Ellerhorst J, Troncoso P, Xu XC et al (1999) Galectin-1 and galectin-3 expression in human prostate tissue and prostate cancer. Urol Res 27(5):362–367
Ellerhorst JA, Stephens LC, Nguyen T et al (2002) Effects of galectin-3 expression on growth and tumorigenicity of the prostate cancer cell line LNCaP. Prostate 50(1):64–70
Saraswati S, Block AS, Davidson MK et al (2011) Galectin-3 is a substrate for prostate specific antigen (PSA) in human seminal plasma. Prostate 71(2):197–208
Bartolazzi A, Gasbarri A, Papotti M et al (2001) Application of an immunodiagnostic method for improving preoperative diagnosis of nodular thyroid lesions. Lancet 357(9269):1644–1650
Meyer A, Coinac I, Bogdanova N et al. (2011) Apoptosis gene polymorphisms and risk of prostate cancer: a hospital-based study of German patients treated with brachytherapy. Urol Oncol (in Press)
Block AS, Saraswati S, Lichti CF et al (2011) Co-purification of Mac-2 binding protein with galectin-3 and association with prostasomes in human semen. Prostate 71(7):711–721
Merseburger AS, Kuczyk MA, Serth J et al (2003) Limitations of tissue microarrays in the evaluation of focal alterations of bcl-2 and p53 in whole mount derived prostate tissues. Oncol Rep 10(1):223–228
Vogel UF (2008) The construction of high-density paraffin tissue microarrays with 0.43-mm-diameter paraffin tissue core biopsies is technically feasible. Virchows Arch 453(1):43–46
Merseburger AS, Hennenlotter J, Simon P et al (2005) Cathepsin D expression in renal cell cancer-clinical implications. Eur Urol 48(3):519–526
Deutscher SL, Figueroa SD, Kumar SR (2009) Tumor targeting and SPECT imaging properties of an (111)In-labeled galectin-3 binding peptide in prostate carcinoma. Nucl Med Biol 36(2):137–146
Than TH, Swethadri GK, Wong J et al (2008) Expression of Galectin-3 and Galectin-7 in thyroid malignancy as potential diagnostic indicators. Singapore Med J 49(4):333–338
Kim MK, Sung CO, Do IG et al (2011) Overexpression of Galectin-3 and its clinical significance in ovarian carcinoma. Int J Clin Oncol 16(4):352–358
Vereecken P, Awada A, Suciu S et al (2009) Evaluation of the prognostic significance of serum galectin-3 in American Joint Committee on Cancer stage III and stage IV melanoma patients. Melanoma Res 19(5):316–320
Johnson KD, Glinskii OV, Mossine VV et al (2007) Galectin-3 as a potential therapeutic target in tumors arising from malignant endothelia. Neoplasia 9(8):662–670
Kobayashi T, Shimura T, Yajima T et al (2011) Transient gene silencing of galectin-3 suppresses pancreatic cancer cell migration and invasion through degradation of β-catenin. Int J Cancer 129(12):2775–2786
Barondes SH, Cooper DN, Gitt MA et al (1994) Galectins. Structure and function of a large family of animal lectins. J Biol Chem 269(33):20807–20810
Konstantinov KN, Robbins BA, Liu FT (1996) Galectin-3, a beta-galactoside-binding animal lectin, is a marker of anaplastic large-cell lymphoma. Am J Pathol 148(1):25–30
Inohara H, Akahani S, Koths K et al (1996) Interactions between galectin-3 and Mac-2-binding protein mediate cell–cell adhesion. Cancer Res 56(19):4530–4534
Ahmed H, Banerjee PP, Vasta GR (2007) Differential expression of galectins in normal, benign and malignant prostate epithelial cells: silencing of galectin-3 expression in prostate cancer by its promoter methylation. Biochem Biophys Res Commun 358(1):241–246
van den Brule FA, Waltregny D, Liu FT et al (2000) Alteration of the cytoplasmic/nuclear expression pattern of galectin-3 correlates with prostate carcinoma progression. Int J Cancer 89(4):361–367
Pacis RA, Pilat MJ, Pienta KJ et al (2000) Decreased galectin-3 expression in prostate cancer. Prostate 44(2):118–123
Ahmed H, Cappello F, Rodolico V et al (2009) Evidence of heavy methylation in the galectin 3 promoter in early stages of prostate adenocarcinoma: development and validation of a methylated marker for early diagnosis of prostate cancer. Transl Oncol 2(3):146–156
Acknowledgments
Judith Knapp and Mario Kramer were supported by the International Academy of Life Sciences, Biomedical Science Exchange Program fellowship. Soum D Lokeshwar is a volunteer at the Sylvester Comprehensive Cancer Center.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Judith S. Knapp and Soum D. Lokeshwar contributed equally to the manuscript.
Rights and permissions
About this article
Cite this article
Knapp, J.S., Lokeshwar, S.D., Vogel, U. et al. Galectin-3 expression in prostate cancer and benign prostate tissues: correlation with biochemical recurrence. World J Urol 31, 351–358 (2013). https://doi.org/10.1007/s00345-012-0925-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-012-0925-y