Abstract
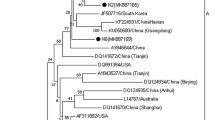
Chicken infectious anemia (CIA), caused by chicken anemia virus (CAV), is an important immunosuppressive disease that seriously threatens the global poultry industry. Here, we isolated and identified 30 new CAV strains from CAV-positive flocks. The VP1 genes of these strains were sequenced and analyzed at the nucleotide and amino acid levels and were found to have very similar nucleotide sequences (> 97% identity); however, they showed 93.9–100.0% sequence identity to the VP1 genes of 55 reference strains. Furthermore, alignment of the deduced amino acid sequences revealed some unique mutations. Phylogenetic analysis indicated the division of VP1 amino acid sequences into two groups (A and B) and four subgroups (A1, A2, A3 and A4). Interestingly, 22 of the newly isolated strains and some Asian reference strains belonged to the A1 group, whereas the remaining eight new isolates belonged to the A3 group. To evaluate the pathogenicity of the epidemic CAV strains from China, the representative strains CAV-JL16/8901 and CAV-HeN19/3001 and the reference strain Cux-1 were selected for animal experiments. Chickens infected with the isolates and reference strain all showed thymus atrophy and bone marrow yellowing. The mortality rates for CAV-JL16/8901, CAV-HeN19/3001, and the reference strain was 30%, 20%, and 0%, respectively, indicating that the epidemic strains pose a more serious threat to chickens. We not only analyzed the molecular evolution of the epidemic strains but also showed for the first time that the epidemic strains in China are more pathogenic than reference strain Cux-1. Effective measures should be established to prevent the spread of CIA in China.






Similar content being viewed by others
References
Rosario K, Breitbart M, Harrach B, Segalés J, Delwart E, Biagini P, Varsani A (2017) Revisiting the taxonomy of the family Circoviridae: establishment of the genus Cyclovirus and removal of the genus Gyrovirus. Arch Virol 162:1447–1463
Todd D, Creelan JL, Mackie DP, Rixon F, McNulty MS (1990) Purification and biochemical characterization of chicken anaemia agent. J Gen Virol 71(Pt 4):819–823
Renshaw RW, Soiné C, Weinkle T, O’Connell PH, Ohashi K, Watson S, Lucio B, Harrington S, Schat KA (1996) A hypervariable region in VP1 of chicken infectious anemia virus mediates rate of spread and cell tropism in tissue culture. J Virol 70:8872–8878
Todd D, McNulty MS, Adair BM, Allan GM (2001) Animal circoviruses. Adv Virus Res 57:1–70
Adair BM (2000) Immunopathogenesis of chicken anemia virus infection. Dev Comp Immunol 24:247–255
Yuasa N, Taniguchi T, Yoshida I (1979) Isolation and some characteristics of an agent inducing anemia in chicks. Avian Dis 23:366–385
Rosenberger JK, Cloud SS (1989) The isolation and characterization of chicken anemia agent (CAA) from broilers in the United States. Avian Dis 33:707–713
Kim HR, Kwon YK, Bae YC, Oem JK, Lee OS (2010) Molecular characterization of chicken infectious anemia viruses detected from breeder and broiler chickens in South Korea. Poult Sci 89:2426–2431
Ducatez MF, Chen H, Guan Y, Muller CP (2008) Molecular epidemiology of chicken anemia virus (CAV) in southeastern Chinese live birds markets. Avian Dis 52:68–73
Olszewska-Tomczyk M, Świętoń E, Minta Z, Śmietanka K (2016) Occurrence and phylogenetic studies of chicken anemia virus from polish broiler flocks. Avian Dis 60:70–74
Zhou W, Yang B, Shen B, Han S, Zhou J (1996) A serologic survey of antibody against chicken infectious anemia virus by indirect immunofluorescent assay in domestic poultry in China. Avian Dis 40:358–360
Zhou W, Shen B, Yang B, Han S, Wei L, Xiao B, Zhou J (1997) Isolation and identification of chicken infectious anemia virus in China. Avian Dis 41:361–364
Li Y, Fang L, Cui S, Fu J, Li X, Zhang H, Cui Z, Chang S, Shi W, Zhao P (2017) Genomic characterization of recent chicken anemia virus isolates in China. Front Microbiol 8:401
Yamaguchi S, Imada T, Kaji N, Mase M, Tsukamoto K, Tanimura N, Yuasa N (2001) Identification of a genetic determinant of pathogenicity in chicken anaemia virus. J Gen Virol 82:1233–1238
Reed LJ (1938) A simple method of examining fifty percent endpoints. Am J Hyg 27:493–497
Schat KA (2009) Chicken anemia virus. Curr Top Microbiol Immunol 331:151–183
Todd D, Connor TJ, Creelan JL, Borghmans BJ, Calvert VM, McNulty MS (1998) Effect of multiple cell culture passages on the biological behaviour of chicken anaemia virus. Avian Pathol 27:74–79
Todd D, Scott AN, Ball NW, Borghmans BJ, Adair BM (2002) Molecular basis of the attenuation exhibited by molecularly cloned highly passaged chicken anemia virus isolates. J Virol 76:8472–8474
Munger J, Hagglund R, Roizman B (2003) Infected cell protein No. 22 is subject to proteolytic cleavage by caspases activated by a mutant that induces apoptosis. Virology 305:364–370
Yao S, Tuo T, Gao X, Han C, Yan N, Liu A, Gao H, Gao Y, Cui H, Liu C, Zhang Y, Qi X, Hussain A, Wang Y, Wang X (2019) Molecular epidemiology of chicken anaemia virus in sick chickens in China from 2014 to 2015. PLoS ONE 14:e0210696
Miller MM, Schat KA (2004) Chicken infectious anemia virus: an example of the ultimate host-parasite relationship. Avian Dis 48:734–745
Yang L, Yixin W, Lichun F, Jiayuan F, Shuai C, Yingjie Z, Zhizhong C, Shuang C, Peng Z (2016) Genomic analysis of the chicken infectious anemia virus in a specific pathogen-free chicken population in China. Biomed Res Int 2016:1–5
Li Y, Hu Y, Cui S, Fu J, Wang Y, Cui Z, Fang L, Chang S, Zhao P (2017) Molecular characterization of chicken infectious anemia virus from contaminated live-virus vaccines. Poult Sci 96:1045–1051
Koch G, van Roozelaar DJ, Verschueren CA, van der Eb AJ, Noteborn MH (1995) Immunogenic and protective properties of chicken anaemia virus proteins expressed by baculovirus. Vaccine 13:763–770
Erfan AM, Selim AA, Naguib MM (2018) Characterization of full genome sequences of chicken anemia viruses circulating in Egypt reveals distinct genetic diversity and evidence of recombination. Virus Res 251:78–85
Marin SY, Barrios PR, Rios RL, Resende M, Resende JS, Santos BM, Martinsa NR (2013) Molecular characterization of contaminating infectious anemia virus of chickens in live commercial vaccines produced in the 1990s. Avian Dis 57:15–21
Zhang X, Liu Y, Wu B, Sun B, Chen F, Ji J, Ma J, Xie Q (2013) Phylogenetic and molecular characterization of chicken anemia virus in southern China from 2011 to 2012. Sci Rep 3:3519
Craig MI, Rimondi A, Delamer M, Sansalone P, König G, Vagnozzi A, Pereda A (2009) Molecular Characterization of Chicken Infectious Anemia Virus Circulating in Argentina During 2007. Avian Dis 4(53):331–333
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG, Thompson JD (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500
Todd D, Creelan JL, Connor TJ, Ball NW, Scott AN, Meehan BM, McKenna GF, McNulty MS (2003) Investigation of the unstable attenuation exhibited by a chicken anaemia virus isolate. Avian Pathol 32:375–382
McKenna GF, Todd D, Borghmans BJ, Welsh MD, Adair BM (2003) Immunopathologic investigations with an attenuated chicken anemia virus in day-old chickens. Avian Dis 47:1339–1345
Natesan S, Kataria JM, Dhama K, Rahul S, Bhardwaj N (2006) Biological and molecular characterization of chicken anaemia virus isolates of Indian origin. Virus Res 118:78–86
Ezzi A, Shoushtari A, Mardjanmehr H (2010) Experimental studies of pathogenecity of chicken infectious anaemia virus (three isolates) in Iran. J Comp Pathol 146:60–65
Acknowledgements
This study was supported by the National Key Research and Development Program of China [Grant number 2016YFE0203200] and the Earmarked Fund for Modern Agriculture Industry Technology Research System [Grant number CARS-41-G15].
Author information
Authors and Affiliations
Contributions
YL, NY, and YZ performed experiments. YL, YW, AL, XL, and BY assembled and analyzed the data and prepared the figures. YZ, YW, CL, and YG contributed to the experimental design and interpretation. HG provided reagents. XW, XQ, HC, KL, LG, and QP provided valuable suggestions. YL, YZ, and XW wrote the paper, and all other authors critically reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Handling Editor: Ana Cristina Bratanich.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Yan, N., Wang, Y. et al. Molecular evolution and pathogenicity of chicken anemia virus isolates in China. Arch Virol 166, 439–449 (2021). https://doi.org/10.1007/s00705-020-04909-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-020-04909-8