Abstract
Purpose
To compare gadoxetic acid alone and combined gadoxetic acid/gadofosveset trisodium-enhanced liver MRI for detection of metastases and differentiation of metastases from haemangiomas.
Methods
Ninety-one patients underwent gadoxetic acid-enhanced liver MRI before and after additional injection of gadofosveset. First, two readers retrospectively identified metastases on gadoxetic acid alone enhanced delayed hepatobiliary phase T1-weighted images together with all other MR images (dynamic images, T2-weighted images, diffusion-weighted images). Second, readers assessed additional T1-weighted images obtained after administration of gadofosveset trisodium. For both interpretations, readers rated lesion conspicuity and confidence in differentiating metastases from haemangiomas. Results were compared using alternative free-response receiver-operating characteristic (AFROC) and conventional ROC methods. Histology and follow-up served as reference standard.
Results
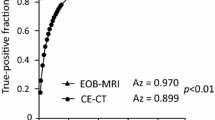
There were 145 metastases and 16 haemangiomas. Both readers detected more metastases using combined gadoxetic acid/gadofosveset (reader 1 = 130; reader 2 = 124) compared to gadoxetic acid alone (reader 1 = 104; reader 2 = 103). Sensitivity of combined gadoxetic acid/gadofosveset (reader 1 = 90 %; reader 2 = 86 %) was higher than that of gadoxetic acid alone (reader 1 = 72 %; reader 2 = 71 %, both P < 0.01). AFROC-AUC was higher for the combined technique (0.92 vs. 0.86, P < 0.001). Sensitivity for correct differentiation of metastases from haemangiomas was higher for the combined technique (reader 1 = 98 %; reader 2 = 99 % vs. reader 1 = 86 %; reader 2 = 91 %, both P < 0.01). ROC-AUC was significantly higher for the combined technique (reader 1 = 1.00; reader 2 = 1.00 vs. reader 1 = 0.87; reader 2 = 0.92, both P < 0.01).
Conclusion
Combined gadoxetic acid/gadofosveset-enhanced MRI improves detection and characterization of liver metastases compared to gadoxetic acid alone.
Key Points
• Combined gadoxetic acid and gadofosveset-enhanced liver MRI significantly improves detection of metastases.
• The combined enhancement technique improves the accuracy to differentiate metastases from haemangiomas.
• Prospective studies need to determine the clinical impact of the combined technique






Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin. doi:10.3322/caac.21262
Fowler KJ, Linehan DC, Menias CO (2013) Colorectal liver metastases: state of the art imaging. Ann Surg Oncol 20:1185–1193
Imam K, Bluemke DA (2000) MR imaging in the evaluation of hepatic metastases. Magn Reson Imaging Clin N Am 8:741–756
Van Beers BE, Pastor CM, Hussain HK (2012) Primovist, Eovist: what to expect? J Hepatol 57:421–429
Huppertz A, Balzer T, Blakeborough A et al (2004) Improved detection of focal liver lesions at MR imaging: multicenter comparison of gadoxetic acid-enhanced MR images with intraoperative findings. Radiology 230:266–275
Kim HJ, Lee SS, Byun JH et al (2014) Incremental value of liver MR imaging in patients with potentially curable colorectal hepatic metastasis detected at CT: a prospective comparison of diffusion-weighted imaging, gadoxetic acid-enhanced MR imaging, and a combination of both MR techniques. Radiology. doi:10.1148/radiol.14140390:140390
Motosugi U, Ichikawa T, Morisaka H et al (2011) Detection of pancreatic carcinoma and liver metastases with gadoxetic acid-enhanced MR imaging: comparison with contrast-enhanced multi-detector row CT. Radiology 260:446–453
Muhi A, Ichikawa T, Motosugi U et al (2011) Diagnosis of colorectal hepatic metastases: comparison of contrast-enhanced CT, contrast-enhanced US, superparamagnetic iron oxide-enhanced MRI, and gadoxetic acid-enhanced MRI. J Magn Reson Imaging 34:326–335
Motosugi U, Ichikawa T, Onohara K et al (2011) Distinguishing hepatic metastasis from hemangioma using gadoxetic acid-enhanced magnetic resonance imaging. Invest Radiol 46:359–365
Goshima S, Kanematsu M, Watanabe H et al (2010) Hepatic hemangioma and metastasis: differentiation with gadoxetate disodium-enhanced 3-T MRI. AJR Am J Roentgenol 195:941–946
Heiken JP (2007) Distinguishing benign from malignant liver tumours. Cancer Imaging 7:S1–S14
Ba-Ssalamah A, Uffmann M, Saini S, Bastati N, Herold C, Schima W (2009) Clinical value of MRI liver-specific contrast agents: a tailored examination for a confident non-invasive diagnosis of focal liver lesions. Eur Radiol 19:342–357
Doo KW, Lee CH, Choi JW, Lee J, Kim KA, Park CM (2009) “Pseudo washout” sign in high-flow hepatic hemangioma on gadoxetic acid contrast-enhanced MRI mimicking hypervascular tumor. AJR Am J Roentgenol 193:W490–W496
Motosugi U, Ichikawa T, Sano K et al (2011) Double-dose gadoxetic acid-enhanced magnetic resonance imaging in patients with chronic liver disease. Invest Radiol 46:141–145
Soyer P, Dufresne AC, Somveille E, Scherrer A (1997) Hepatic cavernous hemangioma: appearance on T2-weighted fast spin-echo MR imaging with and without fat suppression. AJR Am J Roentgenol 168:461–465
Vilgrain V, Boulos L, Vullierme MP, Denys A, Terris B, Menu Y (2000) Imaging of atypical hemangiomas of the liver with pathologic correlation. Radiographics 20:379–397
Higashihara H, Murakami T, Kim T et al (2010) Differential diagnosis between metastatic tumors and nonsolid benign lesions of the liver using ferucarbotran-enhanced MR imaging. Eur J Radiol 73:125–130
Grist TM, Korosec FR, Peters DC et al (1998) Steady-state and dynamic MR angiography with MS-325: initial experience in humans. Radiology 207:539–544
Milot L, Haider M, Foster L, McGregor C, Law C (2012) Gadofosveset trisodium in the investigation of focal liver lesions in noncirrhotic liver: Early experience. J Magn Reson Imaging 36:738–742
Bannas P, Motosugi U, Hernando D, Rahimi MS, Holmes JH, Reeder SB (2015) Combined gadoxetic acid and gadofosveset enhanced liver MRI: a feasibility and parameter optimization study. Magn Reson Med. doi:10.1002/mrm.25554
Itai Y, Furui S, Araki T, Yashiro N, Tasaka A (1980) Computed tomography of cavernous hemangioma of the liver. Radiology 137:149–155
Caseiro-Alves F, Brito J, Araujo AE et al (2007) Liver haemangioma: common and uncommon findings and how to improve the differential diagnosis. Eur Radiol 17:1544–1554
Horton KM, Bluemke DA, Hruban RH, Soyer P, Fishman EK (1999) CT and MR imaging of benign hepatic and biliary tumors. Radiographics 19:431–451
Nagle SK, Busse RF, Brau AC et al (2012) High resolution navigated three-dimensional T(1)-weighted hepatobiliary MRI using gadoxetic acid optimized for 1.5 Tesla. J Magn Reson Imaging 36:890–899
Kuhn JP, Holmes JH, Brau AC, Iwadate Y, Hernando D, Reeder SB (2014) Navigator flip angle optimization for free-breathing T1-weighted hepatobiliary phase imaging with gadoxetic acid. J Magn Reson Imaging 40:1129–1136
Ward J, Naik KS, Guthrie JA, Wilson D, Robinson PJ (1999) Hepatic lesion detection: comparison of MR imaging after the administration of superparamagnetic iron oxide with dual-phase CT by using alternative-free response receiver operating characteristic analysis. Radiology 210:459–466
Chakraborty DP, Berbaum KS (2004) Observer studies involving detection and localization: modeling, analysis, and validation. Med Phys 31:2313–2330
Chakraborty DP, Winter LH (1990) Free-response methodology: alternate analysis and a new observer-performance experiment. Radiology 174:873–881
Liang KY, Zeger SL (1986) Longitudinal data analysis using generalized linear models. Biometrika 73:13–22
Obuchowski NA (1998) On the comparison of correlated proportions for clustered data. Stat Med 17:1495–1507
Jung SH, Kang SH, Ahn C (2003) Chi-square test for R x C contingency tables with clustered data. J Biopharm Stat 13:241–251
Obuchowski NA (1997) Nonparametric analysis of clustered ROC curve data. Biometrics 53:567–578
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
JAFROC Software v. 4.2.1. Available via http://www.devchakraborty.com
Zech CJ, Korpraphong P, Huppertz A et al (2014) Randomized multicentre trial of gadoxetic acid-enhanced MRI versus conventional MRI or CT in the staging of colorectal cancer liver metastases. Br J Surg 101:613–621
Acknowledgments
The scientific guarantor of this publication is Scott B. Reeder. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. Alejandro Munoz del Rio, PhD kindly provided statistical advice for this manuscript. Institutional review board approval was obtained. Written informed consent was waived by the institutional review board. Study subjects or cohorts have not been previously reported. Methodology: retrospective, cross-sectional study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Movie 1
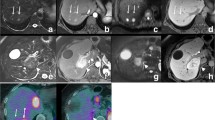
Scrolling through images with gadoxetic acid alone and combined gadoxetic acid/gadofosveset-enhanced liver MRI images provides a much greater appreciation for the look and feel of the improvement in visualization of the combined contrast protocol. Note the improved conspicuity of the small perivascular metastasis in liver segment 4a/b in this 53-year-old woman with oesophageal cancer (same as in Fig. 3b). This histologically confirmed metastases was missed by both readers on gadoxetic alone-enhanced images but detected on combined gadoxetic acid/gadofosveset-enhanced images. (MOV 26107 kb)
Supplementary Movie 2
(MOV 30668 kb)
Rights and permissions
About this article
Cite this article
Bannas, P., Bookwalter, C.A., Ziemlewicz, T. et al. Combined gadoxetic acid and gadofosveset enhanced liver MRI for detection and characterization of liver metastases. Eur Radiol 27, 32–40 (2017). https://doi.org/10.1007/s00330-016-4375-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4375-6