Abstract
Background
Recent reports have described favorable response rates for immune checkpoint inhibitors in prostate cancers with microsatellite instability (MSI). However, it is unclear whether MSI affects the entire tumor mass or is distributed heterogeneously, the latter potentially impairing treatment efficiency.
Methods
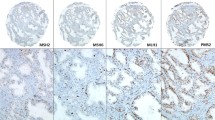
To identify prostate cancers with MSI, 316 advanced prostate cancers were analyzed by immunohistochemistry (IHC) for the mismatch repair (MMR) proteins MLH1, PMS2, MSH2, and MSH6 on a TMA format.
Results
Out of 200 interpretable cancers, IHC findings were consistent with MSI in 10 tumors. In 9 of these 10 cancers, tissue blocks were available for subsequent large section IHC, confirming MSI in 6 cases, each with combined protein loss of MSH2 and MSH6. One additional tumor with unequivocal loss of MLH1 and PMS2 on the TMA, for which further analyses could not be carried out due to lack of tissue, was also considered to exhibit MSI. In total, 7 of 200 interpretable advanced prostate cancers were found to exhibit MMR deficiency/MSI (3.5%). Subsequent analysis of all available cancer-containing archived tissue blocks (n=114) revealed consistent and homogeneous MMR protein loss in each case. Polymerase chain reaction (PCR)-based analysis using the “Bethesda panel” could be executed in 6 MMR deficient tumors of which 4 were MSI-high and 2 were MSI-low.
Conclusions
The absence of intratumoral heterogeneity for the MMR status suggests that MSI occurs early in prostate cancer. It is concluded that MMR analysis on limited biopsy material by IHC is sufficient to estimate the MMR status of the entire cancer mass.


Similar content being viewed by others
References
Abida W, Cheng ML, Armenia J, Middha S, Autio KA, Vargas HA, Rathkopf D, Morris MJ, Danila DC, Slovin SF, Carbone E, Barnett ES, Hullings M, Hechtman JF, Zehir A, Shia J, Jonsson P, Stadler ZK, Srinivasan P, Laudone VP, Reuter V, Wolchok JD, Socci ND, Taylor BS, Berger MF, Kantoff PW, Sawyers CL, Schultz N, Solit DB, Gopalan A, Scher HI (2018) Analysis of the Prevalence of Microsatellite Instability in Prostate Cancer and Response to Immune Checkpoint Blockade. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.5801
Albero-Gonzalez R, Hernandez-Llodra S, Juanpere N, Lorenzo M, Lloret A, Segales L, Duran X, Fumado L, Cecchini L, Lloreta-Trull J (2019) Immunohistochemical expression of mismatch repair proteins (MSH2, MSH6, MLH1, and PMS2) in prostate cancer: correlation with grade groups (WHO 2016) and ERG and PTEN status. Virchows Arch 475:223–231. https://doi.org/10.1007/s00428-019-02591-z
Ashktorab H, Ahuja S, Kannan L, Llor X, Ellis NA, Xicola RM, Laiyemo AO, Carethers JM, Brim H, Nouraie M (2016) A meta-analysis of MSI frequency and race in colorectal cancer. Oncotarget 7:34546–34557. https://doi.org/10.18632/oncotarget.8945
Azzouzi AR, Catto JW, Rehman I, Larre S, Roupret M, Feeley KM, Cussenot O, Meuth M, Hamdy FC (2007) Clinically localised prostate cancer is microsatellite stable. BJU Int 99:1031–1035. https://doi.org/10.1111/j.1464-410X.2006.06723.x
Backes FJ, Leon ME, Ivanov I, Suarez A, Frankel WL, Hampel H, Fowler JM, Copeland LJ, O'Malley DM, Cohn DE (2009) Prospective evaluation of DNA mismatch repair protein expression in primary endometrial cancer. Gynecol Oncol 114:486–490. https://doi.org/10.1016/j.ygyno.2009.05.026
Bartley AN, Luthra R, Saraiya DS, Urbauer DL, Broaddus RR (2012) Identification of cancer patients with Lynch syndrome: clinically significant discordances and problems in tissue-based mismatch repair testing. Cancer Prev Res (Phila) 5:320–327. https://doi.org/10.1158/1940-6207.CAPR-11-0288
Berends MJ, Wu Y, Sijmons RH, Mensink RG, van der Sluis T, Hordijk-Hos JM, de Vries EG, Hollema H, Karrenbeld A, Buys CH, van der Zee AG, Hofstra RM, Kleibeuker JH (2002) Molecular and clinical characteristics of MSH6 variants: an analysis of 25 index carriers of a germline variant. Am J Hum Genet 70:26–37. https://doi.org/10.1086/337944
Boland CR, Thibodeau SN, Hamilton SR, Sidransky D, Eshleman JR, Burt RW, Meltzer SJ, Rodriguez-Bigas MA, Fodde R, Ranzani GN, Srivastava S (1998) A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res 58:5248–5257
Bonneville R, Krook MA, Kautto EA, Miya J, Wing MR, Chen HZ, Reeser JW, Yu L, Roychowdhury S (2017) Landscape of Microsatellite Instability Across 39 Cancer Types. JCO Precis Oncol 2017. https://doi.org/10.1200/PO.17.00073
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Cancer Genome Atlas Research N (2014) Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513:202–209. https://doi.org/10.1038/nature13480
Cancer Genome Atlas Research N (2015) The Molecular Taxonomy of Primary Prostate. Cancer Cell 163:1011–1025. https://doi.org/10.1016/j.cell.2015.10.025
Chapusot C, Martin L, Bouvier AM, Bonithon-Kopp C, Ecarnot-Laubriet A, Rageot D, Ponnelle T, Laurent Puig P, Faivre J, Piard F (2002) Microsatellite instability and intratumoural heterogeneity in 100 right-sided sporadic colon carcinomas. Br J Cancer 87:400–404. https://doi.org/10.1038/sj.bjc.6600474
Cunningham JM, Christensen ER, Tester DJ, Kim CY, Roche PC, Burgart LJ, Thibodeau SN (1998) Hypermethylation of the hMLH1 promoter in colon cancer with microsatellite instability. Cancer Res 58:3455–3460
Dahiya R, Lee C, McCarville J, Hu W, Kaur G, Deng G (1997) High frequency of genetic instability of microsatellites in human prostatic adenocarcinoma. Int J Cancer 72:762–767. https://doi.org/10.1002/(sici)1097-0215(19970904)72:5<762::aid-ijc10>3.0.co;2-b
de Leeuw WJ, Dierssen J, Vasen HF, Wijnen JT, Kenter GG, Meijers-Heijboer H, Brocker-Vriends A, Stormorken A, Moller P, Menko F, Cornelisse CJ, Morreau H (2000) Prediction of a mismatch repair gene defect by microsatellite instability and immunohistochemical analysis in endometrial tumours from HNPCC patients. J Pathol 192:328–335. https://doi.org/10.1002/1096-9896(2000)9999:9999<::AID-PATH701>3.0.CO;2-2
Engel C, Forberg J, Holinski-Feder E, Pagenstecher C, Plaschke J, Kloor M, Poremba C, Pox CP, Ruschoff J, Keller G, Dietmaier W, Rummele P, Friedrichs N, Mangold E, Buettner R, Schackert HK, Kienle P, Stemmler S, Moeslein G, Loeffler M, German HC (2006) Novel strategy for optimal sequential application of clinical criteria, immunohistochemistry and microsatellite analysis in the diagnosis of hereditary nonpolyposis colorectal cancer. Int J Cancer 118:115–122. https://doi.org/10.1002/ijc.21313
Goodfellow PJ, Billingsley CC, Lankes HA, Ali S, Cohn DE, Broaddus RJ, Ramirez N, Pritchard CC, Hampel H, Chassen AS, Simmons LV, Schmidt AP, Gao F, Brinton LA, Backes F, Landrum LM, Geller MA, DiSilvestro PA, Pearl ML, Lele SB, Powell MA, Zaino RJ, Mutch D (2015) Combined Microsatellite Instability, MLH1 Methylation Analysis, and Immunohistochemistry for Lynch Syndrome Screening in Endometrial Cancers From GOG210: An NRG Oncology and Gynecologic Oncology Group Study. J Clin Oncol 33:4301–4308. https://doi.org/10.1200/JCO.2015.63.9518
Grasso CS, Wu YM, Robinson DR, Cao X, Dhanasekaran SM, Khan AP, Quist MJ, Jing X, Lonigro RJ, Brenner JC, Asangani IA, Ateeq B, Chun SY, Siddiqui J, Sam L, Anstett M, Mehra R, Prensner JR, Palanisamy N, Ryslik GA, Vandin F, Raphael BJ, Kunju LP, Rhodes DR, Pienta KJ, Chinnaiyan AM, Tomlins SA (2012) The mutational landscape of lethal castration-resistant prostate cancer. Nature 487:239–243. https://doi.org/10.1038/nature11125
Guedes LB, Antonarakis ES, Schweizer MT, Mirkheshti N, Almutairi F, Park JC, Glavaris S, Hicks J, Eisenberger MA, De Marzo AM, Epstein JI, Isaacs WB, Eshleman JR, Pritchard CC, Lotan TL (2017) MSH2 Loss in Primary Prostate Cancer. Clin Cancer Res 23:6863–6874. https://doi.org/10.1158/1078-0432.CCR-17-0955
Haraldsdottir S, Hampel H, Tomsic J, Frankel WL, Pearlman R, de la Chapelle A, Pritchard CC (2014) Colon and endometrial cancers with mismatch repair deficiency can arise from somatic, rather than germline, mutations. Gastroenterology 147(1308-1316):e1301. https://doi.org/10.1053/j.gastro.2014.08.041
Hempelmann JA, Lockwood CM, Konnick EQ, Schweizer MT, Antonarakis ES, Lotan TL, Montgomery B, Nelson PS, Klemfuss N, Salipante SJ, Pritchard CC (2018) Microsatellite instability in prostate cancer by PCR or next-generation sequencing. J Immunother Cancer 6:29. https://doi.org/10.1186/s40425-018-0341-y
Hinsch A, Brolund M, Hube-Magg C, Kluth M, Simon R, Moller-Koop C, Sauter G, Steurer S, Luebke A, Angerer A, Wittmer C, Neubauer E, Gobel C, Buscheck F, Minner S, Wilczak W, Schlomm T, Jacobsen F, Clauditz TS, Krech T, Tsourlakis MC, Schroeder C (2018) Immunohistochemically detected IDH1(R132H) mutation is rare and mostly heterogeneous in prostate cancer. World J Urol 36:877–882. https://doi.org/10.1007/s00345-018-2225-7
Joost P, Veurink N, Holck S, Klarskov L, Bojesen A, Harbo M, Baldetorp B, Rambech E, Nilbert M (2014) Heterogenous mismatch-repair status in colorectal cancer. Diagn Pathol 9:126. https://doi.org/10.1186/1746-1596-9-126
Kim JY, Shin NR, Kim A, Lee HJ, Park WY, Kim JY, Lee CH, Huh GY, Park DY (2013) Microsatellite instability status in gastric cancer: a reappraisal of its clinical significance and relationship with mucin phenotypes. Korean J Pathol 47:28–35. https://doi.org/10.4132/KoreanJPathol.2013.47.1.28
Kluth M, Meyer D, Krohn A, Freudenthaler F, Bauer M, Salomon G, Heinzer H, Michl U, Steurer S, Simon R, Sauter G, Schlomm T, Minner S (2016) Heterogeneity and chronology of 6q15 deletion and ERG-fusion in prostate cancer. Oncotarget 7:3897–3904. https://doi.org/10.18632/oncotarget.6597
Kluth M, Volta H, Hussein M, Taskin B, Frogh S, Moller-Koop C, Buscheck F, Jacobsen F, Tsourlakis MC, Lubke AM, Hinsch A, Clauditz T, Graefen M, Heinzer H, Huland H, Minner S, Sauter G, Wilczak W, Schlomm T, Simon R (2018) Deletion of 3p13 is a late event linked to progression of TMPRSS2:ERG fusion prostate cancer Cancer Manag Res. 10:5909–5917. https://doi.org/10.2147/CMAR.S172637
Kluth M, Al Kilani Z, Ozden C, Hussein K, Frogh S, Moller-Koop C, Burandt E, Steurer S, Buscheck F, Jacobsen F, Luebke AM, Minner S, Tsourlakis MC, Hoeflmayer D, Wittmer C, Schlomm T, Sauter G, Simon R, Wilczak W (2019) 5q21 deletion is often heterogeneous in prostate cancer. Genes Chromosom Cancer 58:509–515. https://doi.org/10.1002/gcc.22730
Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP (1998) Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4:844–847
Latham A, Srinivasan P, Kemel Y, Shia J, Bandlamudi C, Mandelker D, Middha S, Hechtman J, Zehir A, Dubard-Gault M, Tran C, Stewart C, Sheehan M, Penson A, DeLair D, Yaeger R, Vijai J, Mukherjee S, Galle J, Dickson MA, Janjigian Y, O'Reilly EM, Segal N, Saltz LB, Reidy-Lagunes D, Varghese AM, Bajorin D, Carlo MI, Cadoo K, Walsh MF, Weiser M, Aguilar JG, Klimstra DS, Diaz LA Jr, Baselga J, Zhang L, Ladanyi M, Hyman DM, Solit DB, Robson ME, Taylor BS, Offit K, Berger MF, Stadler ZK (2019) Microsatellite Instability Is Associated With the Presence of Lynch Syndrome. Pan-Cancer J Clin Oncol 37:286–295. https://doi.org/10.1200/JCO.18.00283
Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, Skora AD, Luber BS, Azad NS, Laheru D, Biedrzycki B, Donehower RC, Zaheer A, Fisher GA, Crocenzi TS, Lee JJ, Duffy SM, Goldberg RM, de la Chapelle A, Koshiji M, Bhaijee F, Huebner T, Hruban RH, Wood LD, Cuka N, Pardoll DM, Papadopoulos N, Kinzler KW, Zhou S, Cornish TC, Taube JM, Anders RA, Eshleman JR, Vogelstein B, Diaz LA Jr (2015) PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N Engl J Med 372:2509–2520. https://doi.org/10.1056/NEJMoa1500596
Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, Lu S, Kemberling H, Wilt C, Luber BS, Wong F, Azad NS, Rucki AA, Laheru D, Donehower R, Zaheer A, Fisher GA, Crocenzi TS, Lee JJ, Greten TF, Duffy AG, Ciombor KK, Eyring AD, Lam BH, Joe A, Kang SP, Holdhoff M, Danilova L, Cope L, Meyer C, Zhou S, Goldberg RM, Armstrong DK, Bever KM, Fader AN, Taube J, Housseau F, Spetzler D, Xiao N, Pardoll DM, Papadopoulos N, Kinzler KW, Eshleman JR, Vogelstein B, Anders RA, Diaz LA Jr (2017) Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 357:409–413. https://doi.org/10.1126/science.aan6733
Mathiak M, Warneke VS, Behrens HM, Haag J, Boger C, Kruger S, Rocken C (2017) Clinicopathologic Characteristics of Microsatellite Instable Gastric Carcinomas Revisited: Urgent Need for Standardization. Appl Immunohistochem Mol Morphol 25:12–24. https://doi.org/10.1097/PAI.0000000000000264
Metcalf AM, Spurdle AB (2014) Endometrial tumour BRAF mutations and MLH1 promoter methylation as predictors of germline mismatch repair gene mutation status: a literature review. Familial Cancer 13:1–12. https://doi.org/10.1007/s10689-013-9671-6
Mills AM, Liou S, Ford JM, Berek JS, Pai RK, Longacre TA (2014) Lynch syndrome screening should be considered for all patients with newly diagnosed endometrial cancer. Am J Surg Pathol 38:1501–1509. https://doi.org/10.1097/PAS.0000000000000321
Moreira L, Balaguer F, Lindor N, de la Chapelle A, Hampel H, Aaltonen LA, Hopper JL, Le Marchand L, Gallinger S, Newcomb PA, Haile R, Thibodeau SN, Gunawardena S, Jenkins MA, Buchanan DD, Potter JD, Baron JA, Ahnen DJ, Moreno V, Andreu M, Ponz de Leon M, Rustgi AK, Castells A, Consortium E (2012) Identification of Lynch syndrome among patients with colorectal cancer. JAMA 308:1555–1565. https://doi.org/10.1001/jama.2012.13088
Pai RK, Plesec TP, Abdul-Karim FW, Yang B, Marquard J, Shadrach B, Roma AR (2015) Abrupt loss of MLH1 and PMS2 expression in endometrial carcinoma: molecular and morphologic analysis of 6 cases. Am J Surg Pathol 39:993–999. https://doi.org/10.1097/PAS.0000000000000415
Perinchery G, Nojima D, Goharderakhshan R, Tanaka Y, Alonzo J, Dahiya R (2000) Microsatellite instability of dinucleotide tandem repeat sequences is higher than trinucleotide, tetranucleotide and pentanucleotide repeat sequences in prostate cancer. Int J Oncol 16:1203–1209. https://doi.org/10.3892/ijo.16.6.1203
Pritchard CC, Morrissey C, Kumar A, Zhang X, Smith C, Coleman I, Salipante SJ, Milbank J, Yu M, Grady WM, Tait JF, Corey E, Vessella RL, Walsh T, Shendure J, Nelson PS (2014) Complex MSH2 and MSH6 mutations in hypermutated microsatellite unstable advanced prostate cancer. Nat Commun 5:4988. https://doi.org/10.1038/ncomms5988
Schuster-Bockler B, Lehner B (2012) Chromatin organization is a major influence on regional mutation rates in human cancer cells. Nature 488:504–507. https://doi.org/10.1038/nature11273
Simon R, Panussis S, Maurer R, Spichtin H, Glatz K, Tapia C, Mirlacher M, Rufle A, Torhorst J, Sauter G (2004) KIT (CD117)-positive breast cancers are infrequent and lack KIT gene mutations. Clin Cancer Res 10:178–183
Supek F, Lehner B (2015) Differential DNA mismatch repair underlies mutation rate variation across the human genome. Nature 521:81–84. https://doi.org/10.1038/nature14173
Tsourlakis MC, Stender A, Quaas A, Kluth M, Wittmer C, Haese A, Graefen M, Steurer S, Simon R, Korbel J, Weischenfeldt J, Huland H, Sauter G, Schlomm T, Minner S (2016) Heterogeneity of ERG expression in prostate cancer: a large section mapping study of entire prostatectomy specimens from 125 patients. BMC Cancer 16:641. https://doi.org/10.1186/s12885-016-2674-6
Umar A, Boland CR, Terdiman JP, Syngal S, de la Chapelle A, Ruschoff J, Fishel R, Lindor NM, Burgart LJ, Hamelin R, Hamilton SR, Hiatt RA, Jass J, Lindblom A, Lynch HT, Peltomaki P, Ramsey SD, Rodriguez-Bigas MA, Vasen HF, Hawk ET, Barrett JC, Freedman AN, Srivastava S (2004) Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 96:261–268. https://doi.org/10.1093/jnci/djh034
Velasco A, Hewitt SM, Albert PS, Hossein M, Rosenberg H, Martinez C, Sagalowsky AI, McConnell JD, Marston W, Leach FS (2002) Differential expression of the mismatch repair gene hMSH2 in malignant prostate tissue is associated with cancer recurrence. Cancer 94:690–699. https://doi.org/10.1002/cncr.10247
Watanabe M, Imai H, Shiraishi T, Shimazaki J, Kotake T, Yatani R (1995) Microsatellite instability in human prostate cancer. Br J Cancer 72:562–564. https://doi.org/10.1038/bjc.1995.374
Watanabe M, Imai H, Kato H, Shiraishi T, Ushijima T, Nagao M, Yatani R (1996) Microsatellite instability in latent prostate cancers. Int J Cancer 69:394–397. https://doi.org/10.1002/(SICI)1097-0215(19961021)69:5<394::AID-IJC7>3.0.CO;2-0
Watkins JC, Nucci MR, Ritterhouse LL, Howitt BE, Sholl LM (2016) Unusual Mismatch Repair Immunohistochemical Patterns in Endometrial Carcinoma. Am J Surg Pathol 40:909–916. https://doi.org/10.1097/PAS.0000000000000663
Watson N, Grieu F, Morris M, Harvey J, Stewart C, Schofield L, Goldblatt J, Iacopetta B (2007) Heterogeneous staining for mismatch repair proteins during population-based prescreening for hereditary nonpolyposis colorectal cancer. J Mol Diagn 9:472–478. https://doi.org/10.2353/jmoldx.2007.060162
Wilczak W, Rashed S, Hube-Magg C, Kluth M, Simon R, Buscheck F, Clauditz TS, Grupp K, Minner S, Tsourlakis MC, Moller-Koop C, Graefen M, Adam M, Haese A, Wittmer C, Sauter G, Izbicki JR, Huland H, Schlomm T, Steurer S, Krech T, Lebok P (2017) Up-regulation of mismatch repair genes MSH6, PMS2 and MLH1 parallels development of genetic instability and is linked to tumor aggressiveness and early PSA recurrence in prostate cancer. Carcinogenesis 38:19–27. https://doi.org/10.1093/carcin/bgw116
Acknowledgements
We are grateful to Melanie Witt, Maren Eisenberg, Gabi Rieck, Sina Dietrich, and Jana Hagemann for excellent technical assistance.
Author information
Authors and Affiliations
Contributions
CF, RS, GS, SS: contributed to conception, design, data collection, data analysis and manuscript writing. DH, KM, DD, FB, AH, EB, TC: conception and design, collection of samples. CF, CHM, GMF, MK: collection and data analysis WW, GS, SS: study supervision
Corresponding author
Ethics declarations
The usage of archived diagnostic left-over tissues for manufacturing of TMAs and their analysis for research purposes as well as patient data analysis has been approved by local laws (HmbKHG, §12,1) and by the local ethics committee (Ethics commission Hamburg, WF-049/09). All work has been carried out in compliance with the Helsinki Declaration.
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Quality in Pathology
Rights and permissions
About this article
Cite this article
Fraune, C., Simon, R., Höflmayer, D. et al. High homogeneity of mismatch repair deficiency in advanced prostate cancer. Virchows Arch 476, 745–752 (2020). https://doi.org/10.1007/s00428-019-02701-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-019-02701-x