Abstract
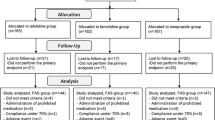
A multicenter, double-blind, randomized controlled trial comparing the efficacy and safety of famotidine with ranitidine in the treatment of acute, benign gastric ulcer disease was coupled with a community-based gastric ulcer disease registry. One hundred ninety-five patients with endoscopically documented gastric ulcer disease were enrolled in the trial and randomly allocated to treatment with either famotidine 40 mg at bedtime or ranitidine 150 mg twice a day. Healing rates were similar in both groups: at four weeks 49% vs 48%, at six weeks 71% vs 69%, and at eight weeks 83% vs 81% for famotidine and for ranitidine, respectively. Pain relief, antacid tablet use, and adverse experiences were also similar in the two groups. Only 25% of patients entered in the gastric ulcer registry were enrolled in the trial. Given that patients with more severe or complicated gastric ulcer disease should be excluded from controlled trials of new drugs, the screening criteria used in the present study resulted in the findings being representative of a quarter of the patients seen in these practices. Therefore, coupling a patient registry with a clinical trial helps determine the applicability of its results. Famotidine 40 mg at bedtime is an effective and well-tolerated treatment of acute, benign gastric ulcer disease and is comparable in efficacy and safety to ranitidine 150 mg twice a day.
Similar content being viewed by others
References
Chremos AN: Pharmacodynamics of Famotidine in Humans. Am J Med 81(suppl 4B):3–7, 1986
Dammann H-G, Muller P, Simon B: 24-hour intragastric acidity and single nighttime dose of three H2-blockers. Lancet 2:1078, 1983
Howard JM, Chremos AN, Collen MJ, McArthur KE, Cherner JA, Maton PN, Ciarleglio CA, Cornelius MJ, Gardner JD, Jensen RT: Famotidine, a new potent, long-acting histamine H2-receptor antagonist: Comparison with cimetidine and ranitidine in the treatment of Zollinger-Ellison syndrome. Gastroenterology 88:1026–1033, 1985
McCallum RW, Chremos AN, Kuljian B, Tupy-Visich MA, Huber PB: MK-208, a novel histamine H2-receptor inhibitor with prolonged antisecretory effects. Dig Dis Sci 30:1139–1144, 1985
Staiger C, Korodnay B, DeVries JX, Weber E, Müller P, Simon B, Dammann H-G: Comparative effects of famotidine and cimetidine on antipyrine kinetics in healthy volunteers. Br J Clin Pharmacol 18:105, 1984
Muller P, Simon B, Dammann H-G, Feurle G, Lichwald K, Schmidt-Gayk H: Acid secretion in man with single daily administration of H2 blockers. Schweiz Med Wochenschr 114:667–671, 1984
Simon B, Muller P, Dammann H-G: Famotidine once-a-day in the therapy of acute, benign gastric ulcer: A worldwide experience. J Clin Gastroenterol 9(suppl 2):19–22, 1987
Paoluzi P, Torsoli A, Bianchi-Porro G, Lazzaroni M, Barbara L, Corinaldesi R, Blasi A, Mangiameli A, Carratelli L, Cheli R, Bovero E, Dal Monte PR, D'Imperio N, Francavilla A, Doronzo F, Mazzacca G, Sabbatini F, Verme G, Ponti V, Speranza V, Lezoche E, Baglioni A, Carotenuto F, Matarazzo PF: Famotidine (MK-208) in the treatment of gastric ulcer. Digestion 32(suppl 1):38–44, 1985
Sackett DL, Gent M: Controversy in counting the attributing events in clinical trials. N Engl J Med 301:1410–1412, 1979
Aitchison J, Bacon-Shone J: Bayesian risk ratio analysis. Am Stat 35:254–257, 1981
Cupples LA, Heeren T, Schatzkin A, Colton T: Multiple testing of hypotheses in comparing two groups. Ann Intern Med 100:122–129, 1984
Hirshowitz BI, DeLuca V, Graham D, Lorber S, Bright-Asare P, Katon R: Treatment of benign chronic gastric ulcer with ranitidine. A randomized, double-blind, and placebocontrolled six-week trial. J Clin Gastroenterol 8:371–376, 1986
Isenberg JI, Peterson WL, Elashoff JD, Sandersfeld MA, Reedy TJ, Ippoliti AF, Van Deventer GM, Frankl H, Longstreth GF, Anderson DS: Healing of benign gastric ulcer with low-dose antacid or cimetidine: A double-blind, randomized, placebo-controlled trial. N Engl J Med 308:1319–1324, 1983
Ashton MG, Holdsworth CD, Ryan FP, Moore M: Healing of gastric ulcers after one, two, and three months of ranitidine. Br Med J 284:467–468, 1982
Dammann H-G, Huttemann W, Kalek HD, Rohner HG, Simon B: Comparative clinical trial of enprostil and ranitidine in the treatment of gastric ulcer. Am J Med 81(suppl 2a):80–84, 1986
Ciclitira PJ, Machell RJ, Farthing MJG, Dick AP, Hunter JO: Double-blind controlled trial of cimetidine in the healing of gastric ulcer. Gut 20:730–734, 1977
Dyck WP, Belsito A, Fleshier B, Liebermann TR, Dickinson PB, Wood JM: Cimetidine and placebo in the treatment of benign gastric ulcer. A multicenter, double-blind study. Gastroenterology 74:410–415, 1978
Hampton JR: Presentation and analysis of the results of clinical trials in cardiovascular disease. Br Med J 282:1371–1373, 1981
The EC/IC Bypass Study Group: Failure of extracranialintracranial arterial bypass to reduce the risk of ischemic stroke. N Engl J Med 313:1191–1200, 1985
Goldring S, Zervas N, Langfitt T: The extracranial-intracranial bypass study. A report of the committee appointed by the American Association of Neurological Surgeons to examine the study. N Engl J Med 316:817–820, 1987
Sundt TM: Was the international randomized trial of extracranial-intracranial arterial bypass representative of the population of risk? N Engl J Med 316:814–816, 1987
Author information
Authors and Affiliations
Additional information
This work was supported, in part, by grants from the National Institutes of Health (AM37199, AM07166, HS04873, and HL17670) and by Merck, Sharp & Dohme Research Laboratories.
Rights and permissions
About this article
Cite this article
Brazer, S.R., Tyor, M.P., Pancotto, F.S. et al. Randomized, double-blind comparison of famotidine with ranitidine in treatment of acute, benign gastric ulcer disease. Digest Dis Sci 34, 1047–1052 (1989). https://doi.org/10.1007/BF01536372
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01536372