Abstract
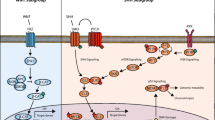
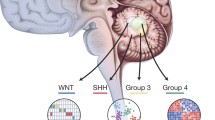
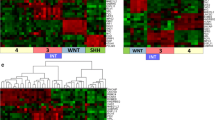
Medulloblastoma, a common pediatric malignant central nervous system tumour, represent a small proportion of brain tumours in adults. Previously it has been shown that in adults, Sonic Hedgehog (SHH)-activated tumours predominate, with Wingless-type (WNT) and Group 4 being less common, but molecular risk stratification remains a challenge. We performed an integrated analysis consisting of genome-wide methylation profiling, copy number profiling, somatic nucleotide variants and correlation of clinical variables across a cohort of 191 adult medulloblastoma cases identified through the Medulloblastoma Advanced Genomics International Consortium. We identified 30 WNT, 112 SHH, 6 Group 3, and 41 Group 4 tumours. Patients with SHH tumours were significantly older at diagnosis compared to other subgroups (p < 0.0001). Five-year progression-free survival (PFS) for WNT, SHH, Group 3, and Group 4 tumours was 64.4 (48.0–86.5), 61.9% (51.6–74.2), 80.0% (95% CI 51.6–100.0), and 44.9% (95% CI 28.6–70.7), respectively (p = 0.06). None of the clinical variables (age, sex, metastatic status, extent of resection, chemotherapy, radiotherapy) were associated with subgroup-specific PFS. Survival among patients with SHH tumours was significantly worse for cases with chromosome 3p loss (HR 2.9, 95% CI 1.1–7.6; p = 0.02), chromosome 10q loss (HR 4.6, 95% CI 2.3–9.4; p < 0.0001), chromosome 17p loss (HR 2.3, 95% CI 1.1–4.8; p = 0.02), and PTCH1 mutations (HR 2.6, 95% CI 1.1–6.2; p = 0.04). The prognostic significance of 3p loss and 10q loss persisted in multivariable regression models. For Group 4 tumours, chromosome 8 loss was strongly associated with improved survival, which was validated in a non-overlapping cohort (combined cohort HR 0.2, 95% CI 0.1–0.7; p = 0.007). Unlike in pediatric medulloblastoma, whole chromosome 11 loss in Group 4 and chromosome 14q loss in SHH was not associated with improved survival, where MYCN, GLI2 and MYC amplification were rare. In sum, we report unique subgroup-specific cytogenetic features of adult medulloblastoma, which are distinct from those in younger patients, and correlate with survival disparities. Our findings suggest that clinical trials that incorporate new strategies tailored to high-risk adult medulloblastoma patients are urgently needed.



Similar content being viewed by others
References
Baroni LV, Sampor C, Gonzalez A, Lubieniecki F, Lamas G, Rugilo C et al (2020) Bridging the treatment gap in infant medulloblastoma: molecularly informed outcomes of a globally feasible regimen. Neuro Oncol 22:1873–1881. https://doi.org/10.1093/neuonc/noaa122
Beier D, Proescholdt M, Reinert C, Pietsch T, Jones DTW, Pfister SM et al (2018) Multicenter pilot study of radiochemotherapy as first-line treatment for adults with medulloblastoma (NOA-07). Neuro Oncol 20:400–410. https://doi.org/10.1093/neuonc/nox155
Brandes AA, Ermani M, Amista P, Basso U, Vastola F, Gardiman M et al (2003) The treatment of adults with medulloblastoma: a prospective study. Int J Radiat Oncol Biol Phys 57:755–761. https://doi.org/10.1016/s0360-3016(03)00643-6
Brandes AA, Franceschi E, Tosoni A, Blatt V, Ermani M (2007) Long-term results of a prospective study on the treatment of medulloblastoma in adults. Cancer 110:2035–2041. https://doi.org/10.1002/cncr.23003
Capper D, Jones DTW, Sill M, Hovestadt V, Schrimpf D, Sturm D et al (2018) DNA methylation-based classification of central nervous system tumours. Nature 555:469–474. https://doi.org/10.1038/nature26000
Cavalli FMG, Remke M, Rampasek L, Peacock J, Shih DJH, Luu B et al (2017) Intertumoral heterogeneity within medulloblastoma subgroups. Cancer Cell 31(737–754):e736. https://doi.org/10.1016/j.ccell.2017.05.005
Franceschi E, Hofer S, Brandes AA, Frappaz D, Kortmann RD, Bromberg J et al (2019) EANO-EURACAN clinical practice guideline for diagnosis, treatment, and follow-up of post-pubertal and adult patients with medulloblastoma. Lancet Oncol 20:e715–e728. https://doi.org/10.1016/s1470-2045(19)30669-2
Franceschi E, Minichillo S, Mura A, Tosoni A, Mascarin M, Tomasello C et al (2020) Adjuvant chemotherapy in average-risk adult medulloblastoma patients improves survival: a long term study. BMC Cancer 20:755. https://doi.org/10.1186/s12885-020-07237-x
Gaviani P, Simonetti G, Rudà R, Franchino F, Lombardi G, Possanzini M et al (2020) Medulloblastoma of the adult: results from a multicenter retrospective study by AINO (Italian Association of Neuro-Oncology) and SIN (Italian Society of Neurology). Neurol Sci. https://doi.org/10.1007/s10072-020-04556-6
Goschzik T, Schwalbe EC, Hicks D, Smith A, Muehlen AZ, Figarella-Branger D et al (2018) Prognostic effect of whole chromosomal aberration signatures in standard-risk, non-WNT/non-SHH medulloblastoma: a retrospective, molecular analysis of the HIT-SIOP PNET 4 trial. Lancet Oncol 19:1602–1616. https://doi.org/10.1016/s1470-2045(18)30532-1
Goschzik T, Muehlen AZ, Doerner E, Waha A, Friedrich C, Hau P et al (2021) Medulloblastoma in adults: cytogenetic phenotypes identify prognostic subgroups. J Neuropathol Exp Neurol 80:419–430. https://doi.org/10.1093/jnen/nlab020
Hovestadt V, Remke M, Kool M, Pietsch T, Northcott PA, Fischer R et al (2013) Robust molecular subgrouping and copy-number profiling of medulloblastoma from small amounts of archival tumour material using high-density DNA methylation arrays. Acta Neuropathol 125:913–916. https://doi.org/10.1007/s00401-013-1126-5
Kann BH, Lester-Coll NH, Park HS, Yeboa DN, Kelly JR, Baehring JM et al (2017) Adjuvant chemotherapy and overall survival in adult medulloblastoma. Neuro Oncol 19:259–269. https://doi.org/10.1093/neuonc/now150
Kocakaya S, Beier CP, Beier D (2016) Chemotherapy increases long-term survival in patients with adult medulloblastoma–a literature-based meta-analysis. Neuro Oncol 18:408–416. https://doi.org/10.1093/neuonc/nov185
Kool M, Jones DT, Jager N, Northcott PA, Pugh TJ, Hovestadt V et al (2014) Genome sequencing of SHH medulloblastoma predicts genotype-related response to smoothened inhibition. Cancer Cell 25:393–405. https://doi.org/10.1016/j.ccr.2014.02.004
Kool M, Korshunov A, Pfister SM (2012) Update on molecular and genetic alterations in adult medulloblastoma. Memo 5:228–232. https://doi.org/10.1007/s12254-012-0037-9
Kool M, Korshunov A, Remke M, Jones DT, Schlanstein M, Northcott PA et al (2012) Molecular subgroups of medulloblastoma: an international meta-analysis of transcriptome, genetic aberrations, and clinical data of WNT, SHH, Group 3, and Group 4 medulloblastomas. Acta Neuropathol 123:473–484. https://doi.org/10.1007/s00401-012-0958-8
Korshunov A, Okonechnikov K, Stichel D, Ryzhova M, Schrimpf D, Sahm F et al (2021) Integrated molecular analysis of adult Sonic Hedgehog (SHH)-activated medulloblastomas reveals two clinically relevant tumor subsets with VEGFA as potent prognostic indicator. Neuro Oncol. https://doi.org/10.1093/neuonc/noab031
Korshunov A, Remke M, Werft W, Benner A, Ryzhova M, Witt H et al (2010) Adult and pediatric medulloblastomas are genetically distinct and require different algorithms for molecular risk stratification. J Clin Oncol 28:3054–3060. https://doi.org/10.1200/jco.2009.25.7121
Lassaletta A, Ramaswamy V (2016) Medulloblastoma in adults: they’re not just big kids. Neuro Oncol 18:895–897. https://doi.org/10.1093/neuonc/now110
Majd N, Penas-Prado M (2019) Updates on management of adult medulloblastoma. Curr Treat Opt Oncol 20:64. https://doi.org/10.1007/s11864-019-0663-0
Nobre L, Zapotocky M, Khan S, Fukuoka K, Fonseca A, McKeown T et al (2020) Pattern of Relapse and treatment response in WNT-activated medulloblastoma. Cell Rep Med. https://doi.org/10.1016/j.xcrm.2020.100038
Nor C, Ramaswamy V (2018) Clinical and pre-clinical utility of genomics in medulloblastoma. Expert Rev Neurother 18:633–647. https://doi.org/10.1080/14737175.2018.1503536
Northcott PA, Buchhalter I, Morrissy AS, Hovestadt V, Weischenfeldt J, Ehrenberger T et al (2017) The whole-genome landscape of medulloblastoma subtypes. Nature 547:311–317. https://doi.org/10.1038/nature22973
Northcott PA, Hielscher T, Dubuc A, Mack S, Shih D, Remke M et al (2011) Pediatric and adult sonic hedgehog medulloblastomas are clinically and molecularly distinct. Acta Neuropathol 122:231–240. https://doi.org/10.1007/s00401-011-0846-7
Ostrom QT, Cioffi G, Gittleman H, Patil N, Waite K, Kruchko C et al (2019) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2012–2016. Neuro Oncol 21:v1–v100. https://doi.org/10.1093/neuonc/noz150
Ramaswamy V, Remke M, Bouffet E, Bailey S, Clifford SC, Doz F et al (2016) Risk stratification of childhood medulloblastoma in the molecular era: the current consensus. Acta Neuropathol 131:821–831. https://doi.org/10.1007/s00401-016-1569-6
Ramaswamy V, Taylor MD (2017) Medulloblastoma: from myth to molecular. J Clin Oncol 35:2355–2363. https://doi.org/10.1200/jco.2017.72.7842
Remke M, Hielscher T, Northcott PA, Witt H, Ryzhova M, Wittmann A et al (2011) Adult medulloblastoma comprises three major molecular variants. J Clin Oncol 29:2717–2723. https://doi.org/10.1200/jco.2011.34.9373
Remke M, Ramaswamy V, Peacock J, Shih DJ, Koelsche C, Northcott PA et al (2013) TERT promoter mutations are highly recurrent in SHH subgroup medulloblastoma. Acta Neuropathol 126:917–929. https://doi.org/10.1007/s00401-013-1198-2
SAS Institute Inc. (2020) Editorial Guidelines https://www.sas.com/en_ca/legal/editorial-guidelines.html. Accessed 02 Apr 2020
Sharma T, Schwalbe EC, Williamson D, Sill M, Hovestadt V, Mynarek M et al (2019) Second-generation molecular subgrouping of medulloblastoma: an international meta-analysis of Group 3 and Group 4 subtypes. Acta Neuropathol 138:309–326. https://doi.org/10.1007/s00401-019-02020-0
Shih DJ, Northcott PA, Remke M, Korshunov A, Ramaswamy V, Kool M et al (2014) Cytogenetic prognostication within medulloblastoma subgroups. J Clin Oncol 32:886–896. https://doi.org/10.1200/JCO.2013.50.9539
Skowron P, Farooq H, Cavalli FMG, Morrissy AS, Ly M, Hendrikse LD et al (2021) The transcriptional landscape of Shh medulloblastoma. Nat Commun 12:1749. https://doi.org/10.1038/s41467-021-21883-0
Sturm D, Witt H, Hovestadt V, Khuong-Quang DA, Jones DT, Konermann C et al (2012) Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell 22:425–437. https://doi.org/10.1016/j.ccr.2012.08.024
Suzuki H, Kumar SA, Shuai S, Diaz-Navarro A, Gutierrez-Fernandez A, De Antonellis P et al (2019) Recurrent noncoding U1 snRNA mutations drive cryptic splicing in SHH medulloblastoma. Nature 574:707–711. https://doi.org/10.1038/s41586-019-1650-0
Wong GC, Li KK, Wang WW, Liu AP, Huang QJ, Chan AK et al (2020) Clinical and mutational profiles of adult medulloblastoma groups. Acta Neuropathol Commun 8:191. https://doi.org/10.1186/s40478-020-01066-6
Zhao F, Ohgaki H, Xu L, Giangaspero F, Li C, Li P, Yang Z, Wang B, Wang X, Wang Z, Ai L, Zhang J, Luo L, Liu P (2016) Molecular subgroups of adult medulloblastoma: a long-term single-institution study. Neuro Oncol 18:982–990. https://doi.org/10.1093/neuonc/now050
Acknowledgements
HC was supported by a Garron Family Cancer Centre Research Fellowship. MDT is supported by operating funds from the National Institutes for Health (5R01CA159859-08 and R01NS106155-01) and the Pediatric Brain Tumor Foundation. V.R. and M.D.T. are supported by a Stand Up To Cancer (SU2C) St. Baldrick’s Pediatric Dream Team translational research grant (SU2C-AACR-DT1113). VR is supported by operating funds from the Canadian Institutes for Health Research, the Canadian Cancer Society Research Institute, the C.R. Younger Foundation, the Garron Family Cancer Centre, Meagan’s Walk, b.r.a.i.n.child and Nelina’s Hope. This study was partially funded with support provided by the Government of Ontario, Ministry of Research, Innovation and Science, and the Princess Margaret Cancer Foundation and support from the Princess Margaret Cancer Centre-OICR Translational Genomics Laboratory. EGVM is supported by operating funds from the National Institutes of Health (R01NS096236 and R01CA235162).
Author information
Authors and Affiliations
Contributions
Experimental design: HC and VR; data collection—all authors; data analysis: HC, LS, KSS, PAN, AK, and VR; interpretation of the data—all authors; funding: VR and MDT; manuscript writing: HC and VR.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
401_2021_2358_MOESM1_ESM.xlsx
Supplementary file1 Online Table 1: Clinical characteristics of 191 adult medulloblastoma patients. Online Table 2: Clinical characteristics of the 25 adult Group 4 validation patients. Online Table 3: Clinical characteristics of the 62 pediatric Group 4 medulloblastoma. (XLSX 33 KB)
Rights and permissions
About this article
Cite this article
Coltin, H., Sundaresan, L., Smith, K.S. et al. Subgroup and subtype-specific outcomes in adult medulloblastoma. Acta Neuropathol 142, 859–871 (2021). https://doi.org/10.1007/s00401-021-02358-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-021-02358-4