Abstract
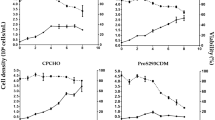
In light of the recent detection of BSE in North America and its endemic nature in other regions of the world, there is a real need to employ cell culture conditions that do not require any animal-derived material. Here we report the use of an ultra-low protein serum-free medium (VP-SFM, Invitrogen) for the amplification of poxviruses in primary chicken embryo fibroblasts (CEF). We compared the amplification of four different poxviruses (canarypox, modified Ankara Virus (MVA), vaccinia virus strain Copenhagen and myxoma strain Lausanne) in three different media: DMEM 10%, DMEM 2% and serum-free medium VP-SFM. VP-SFM is a serum-free, ultra-low protein medium containing no proteins or peptides of human or animal origin designed to support the replication of viruses and the production of recombinant proteins and monoclonal antibodies. Our results show that high titre poxvirus stocks can be prepared in VP-SFM equivalent to that prepared in serum containing medium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Chakrabarti J.R. Sisler B. Moss (1997) ArticleTitleCompactsynthetic, vaccinia virus early/late promoter for protein expression Biotechniques 23 1094–1097 Occurrence Handle1:CAS:528:DyaK2sXnvFCqsLs%3D Occurrence Handle9421642
P.L. Earl N. Cooper L.S. Wyatt B. Moss M.W. Carroll (1998a) Preparation of cell cultures and vaccinia virus stocks F.M. Ausubel R. Brent R.E. Kingston D.D. Moore J.G. Seidman J.A. Smith K. Struhl (Eds) Current Protocols in Molecular Biology Greene Pub. Associates and Wiley Interscience New York 16.16.1–16.16.13
P.L. Earl B. Moss L.S. Wyatt M.W. Carroll (1998b) Generation of recombinant vacinia viruses F.M. Ausubel R. Brent R.E. Kingston D.D. Moore J.G. Seidman J.A. Smith K. Struhl (Eds) Current Protocols in Molecular Biology Greene Pub. Associates and Wiley Interscience New York 16.17.1–16.17.19
R. Fike B. Dadey R. Hassett R. Radominski D. Jayme D. Cady (2001) ArticleTitleAdvanced Granulation Technology (AGTTM). An alternate format for serum-freechemically-defined and protein-free cell culture media Cytotechnology 36 33–39 Occurrence Handle10.1023/A:1014045104525 Occurrence Handle1:CAS:528:DC%2BD38XovV2ksw%3D%3D
C.M. Ford J. Arp T.J. Palker E.E. King G.A. Dekaban (1992) ArticleTitleCharacterization of the antibody response to three different versions of the HTLV-I envelope protein expressed by recombinant vaccinia viruses: induction of neutralizing antibody Virology 191 448–453 Occurrence Handle10.1016/0042-6822(92)90208-7 Occurrence Handle1:CAS:528:DyaK38XmtlWrs70%3D Occurrence Handle1413516
J.B. Griffiths A.J. Racher (1994) ArticleTitleCultural and physiological factors affecting expression of recombinant proteins Cytotechnology 15 3–9 Occurrence Handle10.1007/BF00762374 Occurrence Handle1:STN:280:ByqB3c%2FnsV0%3D Occurrence Handle7765943
J. Hodgson (1991) ArticleTitleChecking sources: the serum supply secret Biotechnology 9 1320–1324 Occurrence Handle1:STN:280:By2C3c3ms1U%3D Occurrence Handle1367762
D.W. Jayme S.R. Smith (2000) ArticleTitleMedia formulation options and manufacturing process controls to safeguard against introduction of animal origin contaminants in animal cell culture Cytotechnology 33 27–36 Occurrence Handle10.1023/A:1008133717035
J.B. Johnston J.W. Barrett W. Chang C.S. Chung W. Zeng J. Masters M. Mann F. Wang J. Cao G. McFadden (2003) ArticleTitleRole of the serine-threonine kinase PAK-1 in myxoma virus replication J. Virol. 77 5877–5888 Occurrence Handle1:CAS:528:DC%2BD3sXjs1Oks7c%3D Occurrence Handle12719581
A. Mayr (1999) ArticleTitleHistorical review of smallpox, the eradication of smallpox and the attenuated smallpox MVA vaccine Berl Munch Tierarztl Wochenschr 112 IssueID9 322–328 Occurrence Handle1:STN:280:DyaK1MvjslSkug%3D%3D Occurrence Handle10507180
O.W. Merten (2002) ArticleTitleDevelopment of serum-free media for cell growth and production of viruses/viral vaccines – safety issues of animal products used in serum-free media Dev. Biol. 111 233–257 Occurrence Handle1:CAS:528:DC%2BD3sXislOgu74%3D
A. Opgenorth K. Graham N. Nation D. Strayer G. McFadden (1992) ArticleTitleDeletion analysis of two tandemly arranged virulence genes in myxoma virus, M11L and myxoma growth factor J. Virol. 66 4720–4731 Occurrence Handle1:CAS:528:DyaK38XmtV2qtL0%3D Occurrence Handle1629952
P. Price E. Evege L. Nestler P. Grefrath F. Naumovic F. Fatunmbi D. Jayme (2002) ArticleTitleA versatile serum-free medium for kidney epithelial cell growth and virus production Focus 19 IssueID3 24–28
Radominski R., Hassett R., Dadey B., Fike R., Cady D. and Jayme D. 2001. Qualification of a novel cell culture medium format. BioPharm 14.
C. Staib G. Sutter (2003) Vaccine protocols: live viral vectors: vaccinia virus A. Robinson M.J. Hudson M.P. Cranage (Eds) Methods in Molecular Medecine Humana Press Totowa NJ 51–68
C. Staib I. Drexler G. Sutter (2004) Vaccinia virus and poxvirology: methods and protocols S.N. Isaacs (Eds) Methods in Molecular Biology Humana Press Towowa NJ 77–99
Staines D. and Price P. 2003. Perspectives in cell culture: managing serum requirements for cell culture. GIBCO Cell Culture.
M. Zang H. Trautmann C. Gandor F. Messi F. Asselbergs C. Leist A. Fiechter J. Reiser (1995) ArticleTitleProduction of recombinant proteins in Chinese hamster ovary cells using a protein-free cell culture medium Biotechnology 13 389–392 Occurrence Handle1:CAS:528:DyaK2MXkslKntrw%3D Occurrence Handle9634782
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gilbert, PA., Comanita, L., Barrett, J. et al. Current Status for High Titre Poxvirus Stock Preparation in CEF Under Serum-Free Medium Conditions: Implication for Vaccine Development. Cytotechnology 48, 79–88 (2005). https://doi.org/10.1007/s10616-005-3795-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10616-005-3795-y