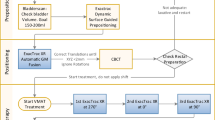
Background and Purpose:
In order to use a single implant with one treatment plan in fractionated high-dose-rate brachytherapy (HDR-B), applicator position shifts must be corrected prior to each fraction. The authors investigated the use of gold markers for X-ray-based setup and position control between the single fractions.
Patients and Methods:
Caudad-cephalad movement of the applicators prior to each HDR-B fraction was determined on radiographs using two to three gold markers, which had been inserted into the prostate as intraprostatic reference, and one to two radiopaque-labeled reference applicators. 35 prostate cancer patients, treated by HDR-B as a monotherapy between 10/2003 and 06/2006 with four fractions of 9.5 Gy each, were analyzed. Toxicity was scored according to the CTCAE Score, version 3.0. Median follow-up was 3 years.
Results:
The mean change of applicators positions compared to baseline varied substantially between HDR-B fractions, being 1.4 mm before fraction 1 (range, –4 to 2 mm), –13.1 mm before fraction 2 (range, –36 to 0 mm), –4.1 mm before fraction 3 (range, –21 to 9 mm), and –2.6 mm at fraction 4 (range, –16 to 9 mm). The original position of the applicators could be readjusted easily prior to each fraction in every patient. In 18 patients (51%), the applicators were at least once readjusted > 10 mm, however, acute or late grade ≥ 2 genitourinary toxicity was not increased (p = 1.0) in these patients.
Conclusion:
Caudad position shifts up to 36 mm were observed. Gold markers represent a valuable tool to ensure setup accuracy and precise dose delivery in fractionated HDR-B monotherapy of prostate cancer.
Hintergrund und Ziel:
Um ein einziges Implantat mit einem Bestrahlungsplan fur die fraktionierte High-Dose-Rate-Brachytherapie (HDR-B) nutzen zu konnen, mussen Positionsverschiebungen der Katheter vor jeder Fraktion erkannt und korrigiert werden. Die Autoren untersuchten den Nutzen von Goldmarkern fur rontgenbildbasierte Konfiguration und Positionskontrolle zwischen den Einzelfraktionen.
Patienten und Methodik:
Die kraniokaudalen Verschiebungen der Applikatoren wurden vor jeder HDR-B-Fraktion anhand von zwei bis drei Goldmarkern als intraprostatische Referenz und ein bis zwei rontgendicht markierten Referenzapplikatoren mittels Rontgenbild bestimmt. 35 Patienten mit Prostatakarzinom, welche zwischen 10/2003 and 06/2006 eine HDR-B als Monotherapie mit vier Fraktionen von jeweils 9,5 Gy erhielten, wurden untersucht. Die Behandlungstoxizitat wurde mit dem CTCAE-Score, Version 3.0, erfasst. Die mediane Nachbeobachtungszeit lag bei 3 Jahren.
Ergebnisse:
Die mittlere Positionsabweichung der Applikatoren von der Sollposition variierte erheblich zwischen den HDR-B-Fraktionen und betrug 1,4 mm vor der ersten Fraktion (Spannweite: –4 bis 2 mm), –13,1 mm vor der zweiten Fraktion (Spannweite: –36 bis 0 mm), –4,1 mm vor der dritten Fraktion (Spannweite: –21 bis 9 mm) und –2,6 mm vor der vierten Fraktion (Spannweite: –16 bis 9 mm). Die ursprungliche Position der Applikatoren konnte bei jedem Patienten problemlos vor jeder Fraktion wiederhergestellt werden. Bei 18 Patienten (51%) wurden die Applikatoren wenigstens einmal > 10 mm verschoben; dennoch war die genitourethrale Akut- oder Spattoxizitat Grad ≥ 2 bei diesen Patienten nicht erhoht (p = 1,0).
Schlussfolgerung:
Positionsverschiebungen von bis zu 36 mm nach kaudal wurden beobachtet. Goldmarker sind bei der fraktionierten HDR-B-Monotherapie des Prostatakarzinoms von Nutzen, um die akkurate Konfiguration und die prazise Verabreichung der Strahlendosis zu gewahrleisten.
Similar content being viewed by others

References
Aebersold DM, Isaak B, Thalmann G, et al. Applicability and dosimetric impact of ultrasound-based preplanning in high-dose-rate brachytherapy of prostate cancer. Strahlenther Onkol 2004;180:351–357.
Bracarda S, de Cobelli O, Greco C, et al. Cancer of the prostate. Crit Rev Oncol Hematol 2005;56:379–396.
Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys 1999;43:1095–1101.
Corner C, Rojas AM, Bryant L, et al. A phase II study of high-dose-rate afterloading brachytherapy as monotherapy for the treatment of localized prostate cancer. Int J Radiat Oncol Biol Phys 2008;72:441–446.
Demanes DJ, Rodriguez RR, Schour L, et al. High-dose-rate intensity-modulated brachytherapy with external beam radiotherapy for prostate cancer: California endocurietherapy’s 10 year results. Int J Radiat Oncol Biol Phys 2005;61:1306–1316.
Dörr W, Jaal J, Zips D. Prostate cancer: biological dose considerations and constraints in tele- and brachytherapy. Strahlenther Onkol 2007;183:14–15.
Eulau SM, Van Hollebeke L, Cavanagh W, et al. High dose rate iridium 192 brachytherapy in localized prostate cancer: results and toxicity with maximum follow-up of 10 years. Int J Radiat Oncol Biol Phys 2000;48:Suppl:149.abstract.
Fowler JF. The linear-quadratic formula and progress in fractionated radiotherapy. Br J Radiol 1989;62:679–694.
Galalae RM, Martinez A, Mate T, et al. Long-term outcome by risk factors using conformal high-dose-rate brachytherapy (HDR-BT) boost with or without neoadjuvant androgen supression for localized prostate cancer. Int J Radiat Oncol Biol Phys 2004;58:1048–1055.
Ghadjar P, Keller T, Rentsch CA, et al. Toxicity and early treatment outcomes in low- and intermediate-risk prostate cancer managed by high-dose-rate brachytherapy as a monotherapy. Brachytherapy 2009;8:45–51.
Grills IS, Martinez AA, Hollander M, et al. High dose rate brachytherapy as prostate cancer monotherapy reduces toxicity compared to low dose rate palladium seeds. J Urol 2004;171:1098–1104.
Guckenberger M, Flentje M. Intensity-modulated radiotherapy (IMRT) of localized prostate cancer: a review and future perspectives. Strahlenther Onkol 2007;183:57–62.
Hiratsuka J, Jo Y, Yoshida K, et al. Clinical results of combined treatment conformal high-dose-rate iridium-192 brachytherapy and external beam radiotherapy using staging lymphadenectomy for localized prostate cancer. Int J Radiat Oncol Biol Phys 2004;59:684–690.
Hoskin PJ, Brownes PJ, Ostler P, et al. High dose rate afterloading brachytherapy for prostate cancer: catheter and gland movement between fractions. Radiother Oncol 2003;68:285–288.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2006. CA Cancer J Clin 2006;56:106–130.
Martin T, Baltas D, Kurek R, et al. 3-D conformal HDR brachytherapy as monotherapy for localized prostate cancer. A pilot study. Strahlenther Onkol 2004;180:225–232.
Martinez AA, Gustafson G, Gonzalez J, et al. Dose escalation using conformal high-dose-rate brachytherapy improves outcome in unfavorable prostate cancer. Int J Radiat Oncol Biol Phys 2002;53:316–327.
Martinez AA, Pataki I, Edmundson G, et al. Phase II prospective study of the use of conformal high-dose-rate brachytherapy as monotherapy for the treatment of favorable stage prostate cancer: a feasibility report. Int J Radiat Oncol Biol Phys 2001;49:61–69.
Pellizzon AC, Nadalin W, Salvajoli JV, et al. Results of high dose rate afterloading brachytherapy boost to conventional external beam radiation therapy for initial and locally advanced prostate cancer. Radiother Oncol 2003;66:167–172.
Pinkawa M, Gagel B, Piroth MD, et al. Changes of dose delivery distribution within the first month after permanent interstitial brachytherapy for prostate cancer. Strahlenther Onkol 2006;182:525–530.
Rades D, Schwarz R, Todorovic M, et al. Experiences with a new high-dose-rate brachytherapy (HDR-BT) boost technique for T3b prostate cancer. Strahlenther Onkol 2007;183:398–402.
Roach M 3rd. Dose escalated external beam radiotherapy versus neoadjuvant androgen deprivation therapy and conventional dose external beam radiotherapy for clinically localized prostate cancer: do we need both? Strahlenther Onkol 2007;Special issue 2:183:26–28.
Roach M 3rd, Hanks G, Thames H Jr, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix consensus conference. Int J Radiat Oncol Biol Phys 2006;65:965–974.
Sobin LH, Wittekind CH. The prostate. In: Sobin LH, Wittekind CH, eds. TNM classification of malignant tumors. New York: Wiley-Liss, 2002:184–187.
Yoshioka Y, Konishi K, Oh RJ, et al. High-dose-rate brachytherapy without external beam irradiation for locally advanced prostate cancer. Radiother Oncol 2006;80:62–68.
Yoshioka Y, Nose T, Yoshida K, et al. High-dose-rate interstitial brachytherapy as a monotherapy for localized prostate cancer: treatment description and preliminary results of a phase I/II clinical trial. Int J Radiat Oncol Biol Phys 2000;48:675–681.
Yoshioka Y, Nose T, Yoshida K, et al. High-dose-rate brachytherapy as monotherapy for localized prostate cancer: a retrospective analysis with special focus on tolerance and chronic toxicity. Int J Radiat Oncol Biol Phys 2003;56:213–220.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghadjar, P., Gwerder, N., Madlung, A. et al. Use of Gold Markers for Setup in Image-Guided Fractionated High-Dose-Rate Brachytherapy as a Monotherapy for Prostate Cancer. Strahlenther Onkol 185, 731–735 (2009). https://doi.org/10.1007/s00066-009-2007-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-009-2007-7