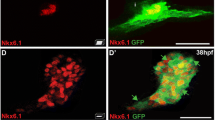
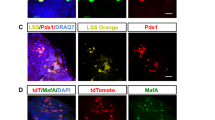
Abstract
Pancreatic β cell clusters produce insulin and play a central role in glucose homeostasis. The regenerative capacity of mammalian β cells is limited and the loss of β cells causes diabetes. In contrast, zebrafish β cell clusters have a high regenerative capacity, making them an attractive model to study β cell cluster regeneration. How zebrafish β cell clusters regenerate, when the regeneration process is complete, and the identification of the cellular source of regeneration are fundamental questions that require investigation. Here, using larval and adult zebrafish, we demonstrate that pancreatic β cell clusters undergo a two-step regeneration process, regenerating functionality and then β cell numbers. Additionally, we found that all regenerating pancreatic β cells arose from Neurod1-expressing cells and that cells from different lineages contribute to both functional and β cell number recovery throughout their life. Furthermore, we found that during development and neogenesis, as well as regeneration, all β cells undergo Neurod1expression in zebrafish. Together, these results shed light on the fundamental cellular mechanisms underlying β cell cluster development, neogenesis, and regeneration.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Andersson O, Adams BA, Yoo D, Ellis GC, Gut P, Anderson RM, German MS, Stainier DYR (2012) Adenosine signaling promotes regeneration of pancreatic β cells in vivo. Cell Metab 6:885–894. https://doi.org/10.1016/j.cmet.2012.04.018
Carril Pardo CA, Massoz L, Dupont MA, Bergemann D, Bourdouxhe J, Lavergne A, Tarifeño-Saldivia E, Helker CS, Stainier DY, Peers B, Voz MM, Manfroid I (2022) A δ-cell subpopulation with a pro-β-cell identity contributes to efficient age-independent recovery in a zebrafish model of diabetes. Elife 11. https://doi.org/10.7554/eLife.67576
Cleaver O, Melton DA (2007) Development of the endocrine pancreas. Joslin’ Dibetes Militus, 14th edn, pp 22–44 (ISBN; 978-0-78-172796-9)
Curado S, Anderson RM, Jungblut B, Mumm J, Schroeter E, Stainier DY (2007) Conditional targeted cell ablation in zebrafish: a new tool for regeneration studies. Dev Dyn 236:1025–1035. https://doi.org/10.1002/dvdy.21100
Delaspre F, Beer RL, Rovira M, Huang W, Wang G, Gee S, Vitery Mdel C, Wheelan SJ, Parsons MJ (2015) Centroacinar Cells Are Progenitors That Contribute to Endocrine Pancreas Regeneration. Diabetes 64:3499–3509. https://doi.org/10.2337/db15-0153
Flasse LC, Pirson JL, Stern DG, Von Berg V, Manfroid I, Peers B, Voz ML (2013) Ascl1b and Neurod1, instead of Neurog3, control pancreatic endocrine cell fate in zebrafish. BMC Biol 11:78. https://doi.org/10.1186/1741-7007-11-78
Ghaye AP, Bergemann D, Tarifeño-Saldivia E, Flasse LC, Von Berg V, Peers B, Voz ML, Manfroid I (2015) Progenitor potential of nkx6.1-expressing cells throughout zebrafish life and during beta cell regeneration. BMC Biol 13:70. https://doi.org/10.1186/s12915-015-0179-4
Gu C, Stein GH, Pan N, Goebbels S, Hörnberg H, Nave KA, Herrera P, White P, Kaestner KH, Sussel L, Lee JE (2010) Pancreatic beta cells require NeuroD to achieve and maintain functional maturity. Cell Metab 11:298–310. https://doi.org/10.1016/j.cmet.2010.03.006
Gut P, Baeza-Raja B, Andersson O, Hasenkamp L, Hsiao J, Hesselson D, Akassoglou K, Verdin E, Hirschey MD, Stainier DY (2013) Whole-organism screening for gluconeogenesis identifies activators of fasting metabolism. Nat Chem Biol 9:97–104. https://doi.org/10.1038/nchembio.1136
Hesselson D, Anderson RM, Stainier DY (2011) Suppression of Ptf1a activity induces acinar-to-endocrine conversion. Curr Biol 21:712–717. https://doi.org/10.1016/j.cub.2011.03.041
Kikuchi K, Gupta V, Wang J, Holdway JE, Wills AA, Fang Y, Poss KD (2011) tcf21+ epicardial cells adopt non-myocardial fates during zebrafish heart development and regeneration. Development 138:2895–2902. https://doi.org/10.1242/dev.067041
Kimmel RA, Onder L, Wilfinger A, Ellertsdottir E, Meyer D (2011) Requirement for Pdx1 in specification of latent endocrine progenitors in zebrafish. BMC Biol 9:75. https://doi.org/10.1186/1741-7007-9-75
Lu J, Liu KC, Schulz N, Karampelias C, Charbord J, Hilding A, Rautio L, Bertolino P, Östenson CG, Brismar K, Andersson O (2016) IGFBP1 increases β-cell regeneration by promoting α- to β-cell transdifferentiation. Embo J 35:2026–2044. https://doi.org/10.15252/embj.201592903
Matsuda H, Parsons MJ, Leach SD (2013) Aldh1-expressing endocrine progenitor cells regulate secondary islet formation in larval zebrafish pancreas. PLoS One 8:e74350. https://doi.org/10.1371/journal.pone.0074350
Matsuda H, Mullapudi ST, Zhang Y, Hesselson D, Stainier DYR (2017) Thyroid Hormone Coordinates Pancreatic Islet Maturation During the Zebrafish Larval-to-Juvenile Transition to Maintain Glucose Homeostasis. Diabetes 66:2623–2635. https://doi.org/10.2337/db16-1476
Matsuda H (2018) Zebrafish as a model for studying functional pancreatic β cells development and regeneration. Dev Growth Differ 60:393–399. https://doi.org/10.1111/dgd.12565
Matsuda H, Mullapudi ST, Yang YHC, Masaki H, Hesselson D, Stainier DYR (2018) Whole-Organism Chemical Screening Identifies Modulators of Pancreatic β-Cell Function. Diabetes 67:2268–2279. https://doi.org/10.2337/db17-1223
McGraw HF, Snelson CD, Prendergast A, Suli A, Raible DW (2012) Postembryonic neuronal addition in zebrafish dorsal root ganglia is regulated by Notch signaling. Neural Dev 7:23. https://doi.org/10.1186/1749-8104-7-23
McMenamin SK, Bain EJ, McCann AE, Patterson LB, Eom DS, Waller ZP, Hamill JC, Kuhlman JA, Eisen JS, Parichy DM (2014) Thyroid hormone-dependent adult pigment cell lineage and pattern in zebrafish. Science 345:1358–1361. https://doi.org/10.1126/science.1256251
Moss JB, Koustubhan P, Greenman M, Parsons MJ, Walter I, Moss LG (2009) Regeneration of the pancreas in adult zebrafish. Diabetes 58:1844–1851. https://doi.org/10.2337/db08-0628
Mullapudi ST, Helker CS, Boezio GL, Maischein HM, Sokol AM, Guenther S, Matsuda H, Kubicek S, Graumann J, Yang YHC, Stainier DY (2018) Screening for insulin-independent pathways that modulate glucose homeostasis identifies androgen receptor antagonists. Elife 7. https://doi.org/10.7554/eLife.42209
Mullapudi ST, Boezio GLM, Rossi A, Marass M, Matsuoka RL, Matsuda H, Helker CSM, Yang YHC, Stainier DYR (2019) Disruption of the pancreatic vasculature in zebrafish affects islet architecture and function. Development 146. https://doi.org/10.1242/dev.173674
Naya FJ, Huang HP, Qiu Y, Mutoh H, DeMayo FJ, Leiter AB, Tsai MJ (1997) Diabetes, defective pancreatic morphogenesis, and abnormal enteroendocrine differentiation in BETA2/neuroD-deficient mice. Genes Dev 11:2323–2334. https://doi.org/10.1101/gad.11.18.2323
Ninov N, Borius M, Stainier DY (2012) Different levels of Notch signaling regulate quiescence, renewal and differentiation in pancreatic endocrine progenitors. Development 139:1557–1567. https://doi.org/10.1242/dev.076000
Obholzer N, Wolfson S, Trapani JG, Mo W, Nechiporuk A, Busch-Nentwich E, Seiler C, Sidi S, Söllner C, Duncan RN, Boehland A, Nicolson T (2008) Vesicular glutamate transporter 3 is required for synaptic transmission in zebrafish hair cells. J Neurosci 28:2110–2118. https://doi.org/10.1523/jneurosci.5230-07.2008
Parsons MJ, Pisharath H, Yusuff S, Moore JC, Siekmann AF, Lawson N, Leach SD (2009) Notch-responsive cells initiate the secondary transition in larval zebrafish pancreas. Mech Dev 126:898–912. https://doi.org/10.1016/j.mod.2009.07.002
Pisharath H, Rhee JM, Swanson MA, Leach SD, Parsons MJ (2007) Targeted ablation of beta cells in the embryonic zebrafish pancreas using E. coli nitroreductase. Mech Dev 124:218–229. https://doi.org/10.1016/j.mod.2006.11.005
Singh SP, Chawla P, Hnatiuk A, Kamel M, Silva LD, Spanjaard B, Eski SE, Janjuha S, Olivares-Chauvet P, Kayisoglu O, Rost F, Bläsche J, Kränkel A, Petzold A, Kurth T, Reinhardt S, Junker JP, Ninov N (2022) A single-cell atlas of de novo β-cell regeneration reveals the contribution of hybrid β/δ-cells to diabetes recovery in zebrafish. Development 149. https://doi.org/10.1242/dev.199853
Thermes V, Grabher C, Ristoratore F, Bourrat F, Choulika A, Wittbrodt J, Joly JS (2002) I-SceI meganuclease mediates highly efficient transgenesis in fish. Mech Dev 118:91–98. https://doi.org/10.1016/s0925-4773(02)00218-6
Tritschler S, Theis FJ, Lickert H, Böttcher A (2017) Systematic single-cell analysis provides new insights into heterogeneity and plasticity of the pancreas. Mol Metab 6:974–990. https://doi.org/10.1016/j.molmet.2017.06.021
Wang H, Wei X, Shi W, He J, Luo L (2020) Key Developmental Regulators Suggest Multiple Origins of Pancreatic Beta Cell Regeneration. Zebrafish. https://doi.org/10.1089/zeb.2019.1777
Wang Y, Rovira M, Yusuff S, Parsons MJ (2011) Genetic inducible fate mapping in larval zebrafish reveals origins of adult insulin-producing β-cells. Development 138:609–617. https://doi.org/10.1242/dev.059097
Ye L, Robertson MA, Hesselson D, Stainier DY, Anderson RM (2015) Glucagon is essential for alpha cell transdifferentiation and beta cell neogenesis. Development 142:1407–1417. https://doi.org/10.1242/dev.117911
Zecchin E, Filippi A, Biemar F, Tiso N, Pauls S, Ellertsdottir E, Gnügge L, Bortolussi M, Driever W, Argenton F (2007) Distinct delta and jagged genes control sequential segregation of pancreatic cell types from precursor pools in zebrafish. Dev Biol 301:192–204. https://doi.org/10.1016/j.ydbio.2006.09.041
Acknowledgements
The authors thank Dr. Didier Stainier (Max Planck Institute for Heart and Lung Research) for the gcga:GFP, ins:Switch, ins:NTR, ins:H2BGFP, and neurod1:EGFP lines, and Dr. David Raible (University of Washington) for plasmids of the neurod1 promoter. RIKEN WT was provided by National Bio Resource Project Zebrafish.
Funding
This work was supported by JSPS KAKENHI Grant Number JP20K06660, the Naito Foundation, and funds from the Ritsumeikan University to HM.
Author information
Authors and Affiliations
Contributions
HM conceived, designed, and performed the experiments and analyzed the data. HM and YK contributed reagents, materials, analysis, and wrote the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplement Fig. 1
ins:Switch (ins:loxp-mCherry-loxp-H2BGFP) systems for expression analysis and lineage tracing. a. Schematic representation of construct of ins:Switch (ins:loxp-mCherry-loxp-H2BGFP). b. Changes in phenotypes of ins:Switch transgenic line with or without Cre or CreERT2. Only mCherryis expressed inthepancreatic β cells of the ins:Switch transgenic line without Cre expression. However, H2BGFP begins to be expressed in β cells after Cre recombination. We always confirmed that there were no leaks of H2BGFP expression without Cre expression using siblings of neurod1:Cre transgenic lines. In the neurod1:CreERT2 transgenic lines, we always confirmed that there were no leaks of H2BGFP expression without 4OHT using siblings of neurod1:CreERT2 transgenic lines used for 4OHT treatment. Scale bars, 40 μm (TIF 16668 KB)
Supplement Fig. 2
Serial z-stack images in gcga:GFP/ins:Switch/ins:NTR. Here, we showed serial z-stack images, which have 6 μm intervals, in a gcga:GFP/ins:Switch/ins:NTR transgenic line at 7 dpa (3 dpa). We found that regenerating β cells (red cells) were always located adjacent to α cells (green cells). Scale bars, 40 μm (TIF 16671 KB)
Supplement Fig. 3
Neurod1 expression pattern in the principal islet in larvae during development and β cell regeneration, using neurod1:GFP/ins:Switch/ins:NTRtreated with or without Mtz. We can see that many neurod1-expressing cells (green cells) existed within the islets during the pancreatic developmental and β cell regenerative processes. Scale bars, 40 μm (TIF 16667 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matsuda, H., Kubota, Y. Zebrafish pancreatic β cell clusters undergo stepwise regeneration using Neurod1-expressing cells from different cell lineages. Cell Tissue Res 394, 131–144 (2023). https://doi.org/10.1007/s00441-023-03805-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-023-03805-2