Abstract
Aims/hypothesis
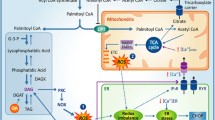
Saturated fatty acids (SFAs) such as palmitate activate inflammatory pathways and elicit an endoplasmic reticulum (ER) stress response in macrophages, thereby contributing to the development of insulin resistance linked to the metabolic syndrome. This study addressed the question of whether or not mitochondrial fatty acid β-oxidation (FAO) affects macrophage responses to SFA.
Methods
We modulated the activity of carnitine palmitoyl transferase 1A (CPT1A) in macrophage-differentiated THP-1 monocytic cells using genetic or pharmacological approaches, treated the cells with palmitate and analysed the proinflammatory and ER stress signatures.
Results
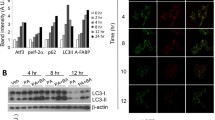
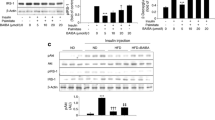
To inhibit FAO, we created THP-1 cells with a stable knockdown (KD) of CPT1A and differentiated them to macrophages. Consequently, in CPT1A-silenced cells FAO was reduced. CPT1A KD in THP-1 macrophages increased proinflammatory signalling, cytokine expression and ER stress responses after palmitate treatment. In addition, in human primary macrophages CPT1A KD elevated palmitate-induced inflammatory gene expression. Pharmacological inhibition of FAO with etomoxir recapitulated the CPT1A KD phenotype. Conversely, overexpression of a malonyl-CoA-insensitive CPT1A M593S mutant reduced inflammatory and ER stress responses to palmitate in THP-1 macrophages. Macrophages with a CPT1A KD accumulated diacylglycerols and triacylglycerols after palmitate treatment, while ceramide accumulation remained unaltered. Moreover, lipidomic analysis of ER phospholipids revealed increased palmitate incorporation into phosphatidylethanolamine and phosphatidylserine classes associated with the CPT1A KD.
Conclusions/interpretation
Our data indicate that FAO attenuates inflammatory and ER stress responses in SFA-exposed macrophages, suggesting an anti-inflammatory impact of drugs that activate FAO.






Similar content being viewed by others
Abbreviations
- AMPK:
-
AMP-dependent protein kinase
- CPT1:
-
Carnitine palmitoyl transferase 1
- CPT1A:
-
Liver isoform of carnitine palmitoyl transferase 1
- CV:
-
Control virus
- DAG:
-
Diacylglycerol
- DFPU:
-
Difluorophenylurethane
- eIF2α:
-
Eukaryotic initiation factor 2α
- ER:
-
Endoplasmic reticulum
- FAO:
-
Fatty acid β-oxidation
- IRE1α:
-
Inositol requiring enzyme 1α
- IκB:
-
Inhibitor of κB
- KD:
-
Knockdown
- LC–MS/MS:
-
Liquid chromatography–tandem mass spectrometry
- MAPK:
-
Mitogen-activated protein kinase
- NLS:
-
Neutral loss scan
- OCR:
-
Oxygen consumption rate
- PBA:
-
Phenylbutyric acid
- PC:
-
Phosphatidylcholine
- PE:
-
Phosphatidylethanolamine
- PEMT:
-
Phosphatidylethanolamine-methyltransferase
- PERK:
-
Protein kinase RNA-like endoplasmic reticulum kinase
- PIS:
-
Precursor ion scan
- PKC:
-
Protein kinase C
- PMA:
-
Phorbol 12-myristate 13-acetate
- PS:
-
Phosphatidylserine
- SFA:
-
Saturated fatty acid
- shRNA:
-
Small hairpin RNA
- siRNA:
-
Small interfering RNA
- sXBP1:
-
Spliced X-box binding protein 1
- TAG:
-
Triacylglycerol
- TLR:
-
Toll-like receptor
References
Glass CK, Olefsky JM (2012) Inflammation and lipid signaling in the etiology of insulin resistance. Cell Metab 15:635–645
Lumeng CN, Saltiel AR (2011) Inflammatory links between obesity and metabolic disease. J Clin Invest 121:2111–2117
Lee JY, Sohn KH, Rhee SH, Hwang D (2001) Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J Biol Chem 276:16683–16689
Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS (2006) TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 116:3015–3025
Pal D, Dasgupta S, Kundu R et al (2012) Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med 18:1279–1285
Haversen L, Danielsson KN, Fogelstrand L, Wiklund O (2009) Induction of proinflammatory cytokines by long-chain saturated fatty acids in human macrophages. Atherosclerosis 202:382–393
Erbay E, Babaev VR, Mayers JR et al (2009) Reducing endoplasmic reticulum stress through a macrophage lipid chaperone alleviates atherosclerosis. Nat Med 15:1383–1391
Hotamisligil GS (2010) Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 140:900–917
Borradaile NM, Han X, Harp JD, Gale SE, Ory DS, Schaffer JE (2006) Disruption of endoplasmic reticulum structure and integrity in lipotoxic cell death. J Lipid Res 47:2726–2737
Holzer RG, Park EJ, Li N et al (2011) Saturated fatty acids induce c-Src clustering within membrane subdomains, leading to JNK activation. Cell 147:173–184
Wen H, Gris D, Lei Y et al (2011) Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat Immunol 12:408–415
Samuel VT, Shulman GI (2012) Mechanisms for insulin resistance: common threads and missing links. Cell 148:852–871
Chavez JA, Summers SA (2012) A ceramide-centric view of insulin resistance. Cell Metab 15:585–594
Koliwad SK, Streeper RS, Monetti M et al (2010) DGAT1-dependent triacylglycerol storage by macrophages protects mice from diet-induced insulin resistance and inflammation. J Clin Invest 120:756–767
Galic S, Fullerton MD, Schertzer JD et al (2011) Hematopoietic AMPK beta1 reduces mouse adipose tissue macrophage inflammation and insulin resistance in obesity. J Clin Invest 121:4903–4915
Morillas M, Gomez-Puertas P, Bentebibel A et al (2003) Identification of conserved amino acid residues in rat liver carnitine palmitoyltransferase I critical for malonyl-CoA inhibition. Mutation of methionine 593 abolishes malonyl-CoA inhibition. J Biol Chem 278:9058–9063
Sebastian D, Herrero L, Serra D, Asins G, Hegardt FG (2007) CPT I overexpression protects L6E9 muscle cells from fatty acid-induced insulin resistance. Am J Physiol Endocrinol Metab 292:E677–E686
Orellana-Gavalda JM, Herrero L, Malandrino MI et al (2011) Molecular therapy for obesity and diabetes based on a long-term increase in hepatic fatty-acid oxidation. Hepatology 53:821–832
Gao X, Li K, Hui X et al (2011) Carnitine palmitoyltransferase 1A prevents fatty acid-induced adipocyte dysfunction through suppression of c-Jun N-terminal kinase. Biochem J 435:723–732
Monsenego J, Mansouri A, Akkaoui M et al (2012) Enhancing liver mitochondrial fatty acid oxidation capacity in obese mice improves insulin sensitivity independently of hepatic steatosis. J Hepatol 56:632–639
Henique C, Mansouri A, Fumey G et al (2010) Increased mitochondrial fatty acid oxidation is sufficient to protect skeletal muscle cells from palmitate-induced apoptosis. J Biol Chem 285:36818–36827
Roduit R, Nolan C, Alarcon C et al (2004) A role for the malonyl-CoA/long-chain acyl-CoA pathway of lipid signaling in the regulation of insulin secretion in response to both fuel and nonfuel stimuli. Diabetes 53:1007–1019
Schiffmann S, Sandner J, Schmidt R et al (2009) The selective COX-2 inhibitor celecoxib modulates sphingolipid synthesis. J Lipid Res 50:32–40
Leiker TJ, Barkley RM, Murphy RC (2011) Analysis of diacylglycerol molecular species in cellular lipid extracts by normal-phase LC-electrospray mass spectrometry. Int J Mass Spectrom 305:103–109
Li Y, Ge M, Ciani L et al (2004) Enrichment of endoplasmic reticulum with cholesterol inhibits sarcoplasmic-endoplasmic reticulum calcium ATPase-2b activity in parallel with increased order of membrane lipids: implications for depletion of endoplasmic reticulum calcium stores and apoptosis in cholesterol-loaded macrophages. J Biol Chem 279:37030–37039
Ekroos K, Chernushevich IV, Simons K, Shevchenko A (2002) Quantitative profiling of phospholipids by multiple precursor ion scanning on a hybrid quadrupole time-of-flight mass spectrometer. Anal Chem 74:941–949
Heiskanen LA, Suoniemi M, Ta HX, Tarasov K, Ekroos K (2013) Long-term performance and stability of molecular shotgun lipidomic analysis of human plasma samples. Anal Chem 85:8757–8763
Ejsing CS, Duchoslav E, Sampaio J et al (2006) Automated identification and quantification of glycerophospholipid molecular species by multiple precursor ion scanning. Anal Chem 78:6202–6214
Jung HR, Sylvanne T, Koistinen KM, Tarasov K, Kauhanen D, Ekroos K (2011) High throughput quantitative molecular lipidomics. Biochim Biophys Acta 1811:925–934
O'Neill LA, Hardie DG (2013) Metabolism of inflammation limited by AMPK and pseudo-starvation. Nature 493:346–355
Volmer R, van der Ploeg K, Ron D (2013) Membrane lipid saturation activates endoplasmic reticulum unfolded protein response transducers through their transmembrane domains. Proc Natl Acad Sci U S A 110:4628–4633
Wong SW, Kwon MJ, Choi AM, Kim HP, Nakahira K, Hwang DH (2009) Fatty acids modulate Toll-like receptor 4 activation through regulation of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. J Biol Chem 284:27384–27392
Nakakuki M, Kawano H, Notsu T, Imada K (2013) Eicosapentaenoic acid suppresses palmitate-induced cytokine production by modulating long-chain acyl-CoA synthetase 1 expression in human THP-1 macrophages. Atherosclerosis 227:289–296
Saraswathi V, Hasty AH (2009) Inhibition of long-chain acyl coenzyme A synthetases during fatty acid loading induces lipotoxicity in macrophages. Arterioscler Thromb Vasc Biol 29:1937–1943
Anderson EK, Hill AA, Hasty AH (2012) Stearic acid accumulation in macrophages induces toll-like receptor 4/2-independent inflammation leading to endoplasmic reticulum stress-mediated apoptosis. Arterioscler Thromb Vasc Biol 32:1687–1695
Qi X, Mochly-Rosen D (2008) The PKCdelta-Abl complex communicates ER stress to the mitochondria - an essential step in subsequent apoptosis. J Cell Sci 121:804–813
Bezy O, Tran TT, Pihlajamaki J et al (2011) PKCdelta regulates hepatic insulin sensitivity and hepatosteatosis in mice and humans. J Clin Invest 121:2504–2517
Jornayvaz FR, Shulman GI (2012) Diacylglycerol activation of protein kinase Cepsilon and hepatic insulin resistance. Cell Metab 15:574–584
Moffitt JH, Fielding BA, Evershed R, Berstan R, Currie JM, Clark A (2005) Adverse physicochemical properties of tripalmitin in beta cells lead to morphological changes and lipotoxicity in vitro. Diabetologia 48:1819–1829
Holland WL, Bikman BT, Wang LP et al (2011) Lipid-induced insulin resistance mediated by the proinflammatory receptor TLR4 requires saturated fatty acid-induced ceramide biosynthesis in mice. J Clin Invest 121:1858–1870
Vance JE, Tasseva G (2013) Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. Biochim Biophys Acta 1831:543–554
Acknowledgements
We thank I. Elschner (Institute of Biochemistry I, Goethe University Frankfurt, Frankfurt, Germany) for technical assistance. We also thank D. Kauhanen and K. Tarasov (Zora Biosciences Oy, Espoo, Finland) for assistance with the shotgun lipidomics analyses.
Funding
This work was supported by grants from Deutsche Forschungsgemeinschaft (grant no. BR999) and by the Else Kröner Fresenius Foundation (Translational Research Innovation—Pharma [TRIP]). Mass spectrometric measurements of DAGs were supported by a grant (RCM) from the US National Institutes of Health (Lipid Maps, GM006938).
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
DN acquired data, drafted and revised the manuscript. SL acquired data and edited the manuscript. TJL and RCM contributed to DAG analysis and revised the manuscript. KE contributed to the shotgun lipidomics analysis and revised the manuscript. NF and GG contributed to ceramide analysis and revised the manuscript. BB analysed data and revised the manuscript. All of the authors approved the final version of the manuscript. BB is responsible for the integrity of the work as a whole.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dmitry Namgaladze and Sebastian Lips contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Fig. 1
(PDF 115 kb)
ESM Fig. 2
(PDF 105 kb)
ESM Fig. 3
(PDF 346 kb)
ESM Table 1
(PDF 89 kb)
Rights and permissions
About this article
Cite this article
Namgaladze, D., Lips, S., Leiker, T.J. et al. Inhibition of macrophage fatty acid β-oxidation exacerbates palmitate-induced inflammatory and endoplasmic reticulum stress responses. Diabetologia 57, 1067–1077 (2014). https://doi.org/10.1007/s00125-014-3173-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-014-3173-4