Abstract
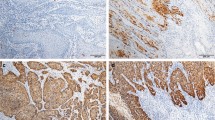
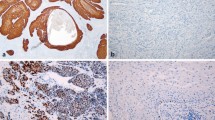
Alterations in the p16/cyclinD1/Rb and ARF/Mdm2/p53 pathways are frequent events in the pathogenesis of squamous cell carcinomas. Different mechanisms of p16 regulation have been described for penile carcinomas so far. Therefore, expression of p16 and p53 was immunohistochemically detected with monoclonal antibodies in 52 primary invasive penile squamous cell carcinomas. The carcinomas were analyzed for allelic loss (LOH) in p16 INK4A and p53, as well as for mutations in the p16 INK4A and the p53 gene. In addition, we examined the promoter status of p16 INK4A by methylation-specific PCR. The presence of human papilloma virus (HPV) 6/11, HPV 16 and HPV 18 DNA was analyzed by PCR. Data were compared to clinical data. Concerning p16, 26 (50%) tumors showed positive immunohistochemistry, 32 (62%) tumors showed allelic loss and 22 tumors (42%) showed promoter hypermethylation. All tumors with negative p16 immunohistochemistry showed LOH near the p16 INK4A locus and/or hypermethylation of the p16 INK4A promoter. HPV 16 DNA was detected in 17 tumors, ten of them with positive p16 immunostaining. The remaining seven tumors with negative p16 staining showed allelic loss and/or promoter hypermethylation. Evidence of lymph node metastasis was significantly associated with negative p16 immunohistochemistry as well as with combined LOH and promoter hypermethylation (p = 0.003 and p = 0.018, respectively). Allelic loss around p53 was found in 22 tumors (42%), and seven mutations of the p53 gene could be demonstrated in our tumors. No correlations could be found between any p53 alteration and clinical parameters.



Similar content being viewed by others
References
Kim WY, Sharpless NE (2006) The regulation of INK4/ARF in cancer and aging. Cell 127:265–275
Sherr CJ (2000) The Pezcoller lecture: cancer cell cycles revisited. Cancer Res 60:3689–3695
zur Hausen H (2009) Papillomaviruses in the causation of human cancers—a brief historical account. Virology 384:260–265
Munoz N, Bosch FX, de Sanjose S, Tafur L, Izarzugaza I, Gili M, Viladiu P, Navarro C, Martos C, Ascunce N et al (1992) The causal link between human papillomavirus and invasive cervical cancer: a population-based case–control study in Colombia and Spain. Int J Cancer 52:743–749
Weinberger PM, Yu Z, Haffty BG, Kowalski D, Harigopal M, Brandsma J, Sasaki C, Joe J, Camp RL, Rimm DL, Psyrri A (2006) Molecular classification identifies a subset of human papillomavirus-associated oropharyngeal cancers with favorable prognosis. J Clin Oncol 24:736–747
Rubin MA, Kleter B, Zhou M, Ayala G, Cubilla AL, Quint WG, Pirog EC (2001) Detection and typing of human papillomavirus DNA in penile carcinoma: evidence for multiple independent pathways of penile carcinogenesis. Am J Pathol 159:1211–1218
Lont AP, Kroon BK, Horenblas S, Gallee MP, Berkhof J, Meijer CJ, Snijders PJ (2006) Presence of high-risk human papillomavirus DNA in penile carcinoma predicts favorable outcome in survival. Int J Cancer 119:1078–1081
Yanagawa N, Osakabe M, Hayashi M, Tamura G, Motoyama T (2008) Detection of HPV-DNA, p53 alterations, and methylation in penile squamous cell carcinoma in Japanese men. Pathol Int 58:477–482
Ferreux E, Lont AP, Horenblas S, Gallee MPW, Raaphorst FM, von Knebel DM, Meijer CJLM, Snijders PJF (2003) Evidence for at least three alternative mechanisms targeting the p16INK4A/Cyclin D/Rb pathway in penile carcinoma, one of which is mediated by high-risk human papillomavirus. J Pathol 201:109–118
Poetsch M, Schuart B-J, Schwesinger G, Kleist B, Protzel C (2007) Screening of microsatellite markers in penile cancer reveals differences between metastatic and non-metastatic carcinomas. Mod Pathol 20:1069–1077
Forbes S, Clements J, Dawson E, Bamford S, Webb T, Dogan A, Flanagan A, Teague J, Wooster R, Futreal PA, Stratton MR (2006) Cosmic 2005. Br J Cancer 94:318–322
Esteller M, Corn PF, Baylin SB, Herman JG (2001) A gene hypermethylation profile of human cancer. Cancer Res 61:3225–3229
Sobin LH, Wittekind C (2002) TNM classification of malignant tumours, 6th edn. Wiley-Liss, New York
Eble JN, Sauter G, Epstein JI, Sesterhenn IA (2004) World Health Organization classification of tumours. Pathology and genetics of tumours of the urinary system and male genital organs. IARC, Lyon, pp 280–290
Hendricksen K, Moonen PM, der Heijden AG, Witjes JA (2006) False-positive lesions detected by fluorescence cystoscopy: any association with p53 and p16 expression? World J Urol 24:597–601
Kong CS, Balzer BL, Troxell ML, Patterson BK, Longacre TA (2007) p16INK4A immunohistochemistry is superior to HPV in situ hybridization for the detection of high-risk HPV in atypical squamous metaplasia. Am J Surg Pathol 31:33–43
Stelow EB, Shaco-Levy R, Bao F, Garcia J, Klimstra DS (2010) Pancreatic acinar cell carcinomas with prominent ductal differentiation: mixed acinar ductal carcinoma and mixed acinar endocrine ductal carcinoma. Am J Surg Pathol 34:510–518
Poetsch M, Dittberner T, Woenckhaus C (2001) PTEN/MMAC1 in malignant melanoma and its importance for tumour progression. Cancer Genet Cytogenet 125:21–26
Poetsch M, Zimmermann A, Wolf E, Kleist B (2005) Loss of heterozygosity occurs predominantly, but not exclusively in the epithelial compartment of pleomorphic adenoma. Neoplasia 7:688–695
Sotlar K, Diemer D, Dethleffs A, Hack Y, Stubner A, Vollmer N, Menton S, Menton M, Dietz K, Wallwiener D, Kandolf R, Bültmann B (2004) Detection and typing of human papillomavirus by E6 nested multiplex PCR. J Clin Microbiol 42:3176–3184
Poetsch M, Lorenz G, Kleist B (2002) Detection of new PTEN/MMAC1 mutations in head and neck squamous cell carcinomas with loss of chromosomes 10. Cancer Genet Cytogenet 132:20–24
Yano M, Hamatani K, Eguchi H, Hirai Y, MacPhee DG, Sugino K, Dohi K, Itamoto T, Asahara T (2007) Prognosis in patients with hepatocellular carcinoma correlates to mutations of p53 and/or hMSH2 genes. Eur J Cancer 43:1092–1100
Liu Y, Lan Q, Siegfried JM, Luketich JD, Keohavong P (2006) Aberrant promoter methylation of p16 and MGMT genes in lung tumors from smoking and never-smoking lung cancer patients. Neoplasia 8:46–51
Prowse DM, Ktori EN, Chandrasekaran D, Prapa A, Baithun S (2008) Human papillomavirus-associated increase in p16INK4A expression in penile lichen sclerosus and squamous cell carcioma. Br J Dermatol 158:261–265
Pascual A, Pariente M, Godinez JM, Sánchez-Prieto R, Atienzar M, Segura M, Poblet E (2007) High prevalence of human papillomavirus 16 in penile carcinoma. Histol Histopathol 22:177–183
Stankiewicz E, Kudahette SC, Prowse DM, Ktori E, Cuzick J, Ambroisine L, Zhang X, Watkin N, Corbishley C, Berney DM (2009) HPV infection and immunochemical detection of cell-cycle markers in verrucous carcinoma of the penis. Mod Pathol 22:1160–1168
Walboomers JM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV, Snijders PF, Peto J, Meijer CJ, Munoz N (1999) Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 189:12–19
Singhi AD, Westra WH (2010) Comparison of human papillomavirus in situ hybridization and p16 immunohistochemistry in the detection of human papillomavirus-associated head and neck cancer based on a prospective clinical experience. Cancer 116:2166–2173
van de Nieuwenhof HP, van Kempen LC, de Hullu JA, Bekkers RL, Bulten J, Melchers WJ, Massuger LF (2009) The etiologic role of HPV in vulvar squamous cell carcinoma fine tuned. Cancer Epidemiol Biomarkers Prev 18:2061–2067
Mulvany NJ, Allen DG, Wilson SM (2008) Diagnostic utility of p16INK4a: a reappraisal of its use in cervical biopsies. Pathology 40:335–344
Galmiche L, Coste-Burel M, Lopez P, Phillippe HJ, Laboisse C (2006) The expression of p16INK4a is not correlated with HPV status in CIN 1. Histopathology 48:767
Senba M, Buziba N, Mori N, Wada A, Irie S, Toriyama K (2009) Detection of human papillomavirus and cellular regulators p16INK4a, p53, and NF.kappaB in penile cancer cases in Kenya. Acta Virol 53:43–48
Smith EM, Rubenstein LM, Hoffman H, Haugen TH, Turek LP (2010) Human papillomavirus, p16 and p53 expression associated with survival of head and neck cancer. Infect Agent Cancer 5:4
Karamitopoulou E, Zlober I, Koumarianou A, Patsouris ES, Peros G, Lugli A (2010) Expression of p16 in lymph node metastases of adjuvantly treated stage III colorectal cancer patients identifies poor prognostic subgroups: a retrospective analysis of biomarkers in matched primary tumor and lymph node metastases. Cancer 116:4474–4486
Schulz P, Scholz A, Rexin A, Hauff P, Schirner M, Wiedenmann B, Detjen K (2008) Inducible re-expression of p16 in an orthotopic mouse model of pancreatic cancer inhibits lymphangiogenesis and lymphatic metastasis. Br J Cancer 99:110–117
Lopes A, Bezerra AL, Pinto CA, Serrano SV, de Mello CA, Villa LL (2002) p53 as a new prognostic factor for lymph node metastasis in penile carcinoma: analysis of 82 patients treated with amputation and bilateral lymphadenectomy. J Urol 168:81–86
Martins AC, Faria SM, Cologna AJ, Suaid HJ, Tucci S Jr (2002) Immunoexpression of p53 protein and proliferating cell nuclear antigen in penile carcinoma. J Urol 167:89–93
Levi JE, Rahal P, Sarkis AS, Villa L (1998) Human papillomarvirus DNA and p53 status in penile carcinomas. Int J Cancer 76:779–783
Gentile V, Vicini P, Giacomelli L, Cardillo MR, Pierangeli A, Degener AM (2006) Detection of human papillomavirus DNA, p53 and ki67 expression in penile carcinomas. Int J Immunopathol Pharmacol 19:209–215
Goebell PJ, Groshen SG, Schmitz-Drager BJ (2010) p53 immunohistochemistry in bladder cancer—a new approach to an old question. Urol Oncol 28:377–388
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poetsch, M., Hemmerich, M., Kakies, C. et al. Alterations in the tumor suppressor gene p16 INK4A are associated with aggressive behavior of penile carcinomas. Virchows Arch 458, 221–229 (2011). https://doi.org/10.1007/s00428-010-1007-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-010-1007-4