Abstract
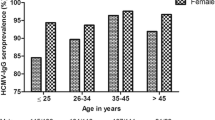
Since the dynamics of transmission of human cytomegalovirus (HCMV) have not been clarified yet, we assessed a possible change in HCMV seroprevalence in Frankfurt am Main, Germany during the past twenty years and tried to detect variables with an impact on epidemiology. Between 1/1/1988 and 10/15/2008, a total of 54443 serum samples were collected for routine diagnostics and analyzed using Enzygnost Anti HCMV-IgG enzyme immunoassay (Siemens/Dade Behring, Marburg, Germany). Two decades, 1/1/1988–12/31/1997 and 1/1/1998–10/15/2008, and several groups (type of health insurance, gender, age, HIV-status) were evaluated to assess changes in seroprevalence. Regarding both decades, the overall age-adjusted prevalence of HIV-negative patients dropped from 63.70% (confidence interval (CI) 95% 63.15–64.25) to 57.25% (CI 95% 56.57–57.93; P < 0.0001). Private health insurance (PHI) patients showed significant lower HCMV seroprevalences than members of obligatory health insurances (OHI) in both decades (1988–1997: PHI = 55.79%, OHI = 64.27%; P < 0.0001; 1998–1908: PHI = 47.02%, OHI = 58.74%; P < 0.0001). Furthermore, comparing the two decades, there was generally a gender-specific statistically significant decrease in HCMV seroprevalence for males (63.54–55.54%) and females (63.83–58.73%) as well as for members of PHI and OHI (PHI males: 57.59% to 47.19%, PHI females 54.10–46.80%; OHI males: 64.00–57.06%, OHI females 64.50–60.11%). Also, while female HIV-positive patients showed significant difference in HCMV seroprevalence between the two decades (83.17 and 87.80%, P = 0.023), there was no significant difference in male patients with HIV (88.76 and 87.32% in the first and second decade, respectively (P = 0.196). The cumulative HCMV prevalence of all HIV-negative patients tested in the past 20 years demonstrates a biphasic, age-related rise of HCMV seroprevalence throughout all age-groups. The seroprevalence of HCMV has declined between 1988–1997 and 1998–2008 in Frankfurt am Main, Germany. The decline varied between different age-groups. HCMV prevalence correlates with the type of health insurance, gender, age, and HIV-status.



Similar content being viewed by others
Abbreviations
- HCMV:
-
Human cytomegalovirus
- CI 95%:
-
95% confidence intervals
- PHI:
-
Private health insurance
- OHI:
-
Obligatory health insurance
References
Campbell AE, Cavanaugh VJ, Slater JS (2008) The salivary glands as a privileged site of cytomegalovirus immune evasion and persistence. Med Microbiol Immunol 197(2):205–213
Doom CM, Hill AB (2008) MHC class I immune evasion in MCMV infection. Med Microbiol Immunol 197(2):191–204
Martin H, Mandron M, Davrinche C (2008) Interplay between human cytomegalovirus and dendritic cells in T cell activation. Med Microbiol Immunol 197(2):179–184
Stinski MF, Isomura H (2008) Role of the cytomegalovirus major immediate early enhancer in acute infection and reactivation from latency. Med Microbiol Immunol 197(2):223–231
Waisman A, Al Croxford, Demircik F (2008) New tools to study the role of B cells in cytomegalovirus infections. Med Microbiol Immunol 197(2):145–149
Waller EC, Day E, Sissons JG, Wills MR (2008) Dynamics of T cell memory in human cytomegalovirus infection. Med Microbiol Immunol 197(2):83–96
Just-Nübling G, Korn S, Ludwig B, Stephan C, Doerr HW, Preiser W (2003) Primary cytomegalovirus infection in an outpatient setting—laboratory markers and clinical aspects. Infection 31(5):318–323
Scholz M, Doerr HW, Cinatl J (2003) Human cytomegalovirus retinitis: pathogenicity, immune evasion and persistence. Trends Microbiol 11(4):171–178
Spector SA (1997) Detection and quantification of human cytomegalovirus (CMV) as a marker for development of CMV disease and survival in patients with AIDS. Antiviral therapy 2(4):200–205
Scholz M, Margraf S, Menon S, Schuller A, Doerr HW, Cinatl J (2004) Supernatants from human cytomegalovirus (HCMV)-infected retinal glial cells increase transepithelial electrical resistance in a cell culture model: evidence of HCMV immune escape in the eye? Med Microbiol Immunol 193(4):205–208
Scholz M, Vogel JU, Höver G, Prösch S, Kotchetkov R, Cintal J, Koch F, Doerr HW, Cinatl J Jr (2004) Thrombin induces Sp1-mediated antiviral effects in cytomegalovirus-infected human retinal pigment epithelial cells. Med Microbiol Immunol 193(4):195–203
Boeckh M, Nichols WG (2003) Immunosuppressive effects of beta-herpes viruses. Herpes 10(1):12–16
Cinatl J Jr, Scholz M, Doerr HW (2005) Role of tumor cell immune escape mechanisms in cytomegalovirus-mediated oncomodulation. Med Res Rev 25(2):167–185
Michaelis M, Doerr HW, Cinatl J (2009) The story of cytomegalovirus and cancer: increasing evidence and open questions. Neoplasia 11(1):1–9
Von Müller L, Mertens T (2008) Human cytomegalovirus infection and antiviral immunity in septic patients without canonical immunosuppression. Med Microbiol Immunol 197(2):75–82
Su BY, Su CY, Yu SF, Chen CJ (2007) Incidental discovery of high systemic lupus erythematosus disease activity associated with cytomegalovirus viral activity. Med Microbiol Immunol 196(3):165–170
Preiser W, Doerr HW, Vogel JU (2003) Virology and epidemiology of oral herpesvirus infections. Med Microbiol Immunol 192(3):133–136
Doerr HW, Lehmair H, Schmitz H, Kampa D, Luthardt T (1977) Simple mathematical deductions in the seroepidemiology of viral infections. I. Herpesvirus group (herpesvirus hominis, varicella-zoster virus, cytomegalovris, Epstein-Barr-Virus). Zentralbl Bakteriol Orig A. 238(2):149–164
Doerr HW (1987) Cytomegalovirus infection in pregnancy. J Virol Methods 17(1–2):127–132
Cannon MJ, Davis KF (2005) Washing our hands of the congenital cytomegalovirus disease epidemic. BMC Public Health 20;5:70
Dollard SC, Grosse SD, Ross DS (2007) New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol 17(5):355–363
Scholz H (2000) Frequency of Congenital cytomegalovirus in Germany; Monatsschr Kinderheilkd 148: 887–890
Kenneson A, Cannon MJ (2007) Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol 17(4):253–276
Pass RF, Fowler KB, Boppana S (1991) Progress in cytomegalovirus research. In: Landini MP (ed) Proceedings of the Third International Cytomegalovirus Workshop Bologna. Italy: June 1991 London: Excerpta Medica 1991, pp 3–10
Pawelec G, Derhovanessian E, Larbi A, Strindhall J, Wikby A (2009) Cytomegalovirus and human immunosenescence. Rev Med Virol 19(1):47–56
Koch S, Solana R, Dela Rosa O, Pawelec G (2006) Human cytomegalovirus infection and T cell immunosenescence: a mini review. Mech Ageing Dev 127(6):538–543
Almeida LN, Azevedo RS, Amaku M, Massad E (2001) Cytomegalovirus seroepidemiology in an urban community of São Paulo, Brazil. Rev Saude Publica 35(2):124–129
Hecker M, Qiu D, Marquardt K, Bein G, Hackstein H (2004) Continuous cytomegalovirus seroconversion in a large group of healthy blood donors. Vox Sang 86(1):41–44
Dowd JB, Aiello AE, Alley DE (2009) Socioeconomic disparities in the seroprevalence of cytomegalovirus infection in the US population: NHANES III. Epidemiol Infect 137(1):58–65
Weber B, Fall EM, Berger A, Doerr HW (1999) Screening of blood donors for human cytomegalovirus (HCMV) IgG antibody with an enzyme immunoassay using recombinant antigens. J Clin Virol 14(3):173–181
De Ory F, Ramírez R, García Comas L, León P, Saqües MJ, Sanz JC (2004) Is there a change in cytomegalovirus seroepidemiology in Spain? Eur J Epidemiol 19(1):85–89
Hoshiba T, Asamoto A, Yabuki Y (1998) Decreasing seropositivity of cytomegalovirus of pregnant women in Japan. Nippon Rinsho. 56(1):193–196
Doerr HW, Rentschler M, Scheifler G (1987) Serologic detection of active infections with human herpesviruses (CMV, EBV, HSV, VZV): diagnostic potential of IgA class and IgG subclass-specific antibodies. Infection. 15(2):93–98
Svahn A, Berggren J, Parke A, Storsaeter J, Thorstensson R, Linde A (2006) Change in seroprevalence to four herpes viruses over 30 years in Swedish children aged 9–12 years. J Clin Virol 37(2):118–123
Preiser W, Fleckenstein C, Doerr HW (2004) New methods for the diagnosis of cytomegalovirus. Dtsch Med Wochenschr 129(27):1509–1512
Staras SA, Dollard SC, Radford KW, Flanders WD, Pass RF, Cannon MJ (2006) Seroprevalence of cytomegalovirus infection in the United States, 1988–1994. Clin Infect Dis 43(9):1143–1151
Gude J (2009) Divorces 2007, Statistisches Bundesamt, Wiesbaden
Doerr HW, Braun R, Munk K (1985) Human cytomegalovirus infection: recent developments in diagnosis and epidemiology. Klin Wochenschr 63:241–251
Enzensberger R, Braun W, July C, Helm EB, Doerr HW (1991) Prevalence of antibodies to human herpesviruses and hepatitis B virus in patients at different stages of human immunodeficiency virus (HIV) infection. Infection. 19(3):140–145
Births in Germany, Statistisches Bundesamt, Wiesbaden 2007; Bestellnummer: 0120007-07900-4
Staras SA, Flanders WD, Dollard SC, Pass RF, McGowan JE Jr, Cannon MJ (2008) Influence of sexual activity on cytomegalovirus seroprevalence in the United States, 1988–1994. Sex Transm Dis 35(5):472–479
Tookey PA, Ades AE, Peckham CS (1992) Cytomegalovirus prevalence in pregnant women: the influence of parity. Arch Dis Child 67(7 Spec No):779–783
Halisch J (2008) Frankfurter Integrationsstudie, efms
Uyar Y, Balci A, Akcali A, Cabar C (2008) Prevalence of rubella and cytomegalovirus antibodies among pregnant women in northern Turkey. New Microbiol 31(4):451–455
Taber LH, Frank AL, Yow MD, Bagley A (1985) Acquisition of cytomegaloviral infections in families with young children: a serological study. J Infect Dis 151(5):948–952
Staras SA, Flanders WD, Dollard SC, Pass RF, McGowan JE Jr, Cannon MJ (2008) Cytomegalovirus seroprevalence and childhood sources of infection: a population-based study among pre-adolescents in the United States. J Clin Virol. 43(3):266–271
Pass RF, Little EA, Stagno S, Britt WJ, Alford CA (1987) Young children as a probable source of maternal and congenital cytomegalovirus infection. N Engl J Med 316(22):1366–1370
Adler SP (1988) Molecular epidemiology of cytomegalovirus: viral transmission among children attending a day care center, their parents, and caretakers. J Pediatr 112(3):366–372
Rabenau HF, Buxbaum S, Preiser W, Weber B, Doerr HW (2002) Seroprevalence of herpes simplex virus types 1 and type 2 in the Frankfurt am Main area, Germany. Med Microbiol Immunol 190(4):153–160
Buxbaum S, Geers M, Gross G, Schöfer H, Rabenau HF, Doerr HW (2003) Epidemiology of herpes simplex virus types 1 and 2 in Germany: what has changed? Med Microbiol Immunol 192(3):177–181
Weber B, Rabenau H, Berger A, Scheuermann EH, Staszewski S, Kreuz W, Scharrer I, Schoeppe W, Doerr HW (1995) Seroprevalence of HCV, HAV, HBV, HDV, HCMV and HIV in high risk groups/Frankfurt a.M., Germany. Zentralbl Bakteriol 282(1):102–112
Acknowledgments
The authors are grateful to Dr. Henry Ogbomo for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lübeck, P.R., Doerr, H.W. & Rabenau, H.F. Epidemiology of Human Cytomegalovirus (HCMV) in an urban region of Germany: what has changed?. Med Microbiol Immunol 199, 53–60 (2010). https://doi.org/10.1007/s00430-009-0136-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-009-0136-3