Abstract
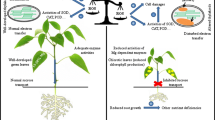
The root/shoot-ratio is a simple parameter to describe the systemic response of plants to alterations of their nutritional status, as indicated by the C/N-balance of leaves. The ‘functional equilibrium hypothesis’ holds that leaf growth is limited by the supply of nitrogen from the roots, whereas root growth depends on the carbon supply from leaves. The nature of the systemic control that balances root and shoot growth is not fully understood. Previous experiments have shown that root growth of transformed tobacco plants, which lack functional root nitrate reductase, was severely impeded, when plants were grown on NO −3 as the sole N-source. In these experiments, the root/shoot-ratio was correlated with the Glutamate/Glutamine-ratio of roots. In the present study we tested the hypothesis that high internal Glu contents (in relation to Gln) inhibit root growth. Wild type and transformed tobacco plants were given access to both NH4 and NO3, and were cultivated at ambient and elevated pCO2 in order to vary carbon availability. The uptake and assimilation of NH +4 by the root was significantly higher in transformed than in wild type tobacco, in particular at elevated pCO2. Consequently, the Glu/Gln-ratio in the root of transformants was significantly lower than in NO −3 -grown plants, and was, in the present study, not different from the wild type. However, we failed to observe a correlation between plant architecture and the Glu/Gln-ratio of roots, suggesting that signals arising from the immediate products of nitrate reduction (nitrite) are involved in the systemic control of root growth. Furthermore the synthesis of root-derived signals, which affect N-turnover, starch re-mobilization and the growth of leaves, appears to be associated with root nitrate reduction. This enzymatic step seems to be indispensable for the systemic control of biomass partitioning, and plays a crucial role for the integration of carbon and nitrogen metabolism at the whole plant level.






Similar content being viewed by others
Abbreviations
- Gln:
-
glutamine
- Glu:
-
glutamate
- LNR:
-
leaf nitrate reductase
- NR:
-
nitrate reductase
References
Andrews M (1986) The partitioning of nitrate assimilation between root and shoot of higher plants. Plant Cell Environ 9:511–519
Andrews M, Lea PJ, Raven JA, Lindsey K (2004) Can genetic manipulation of plant nitrogen assimilation enzymes result in increased crop yield and greater N-use efficiency? An assessment. Ann Appl Biol 145:25–40
Andrews M, Lea PJ, Raven JA, Sprent JI (2006) A role for shoot protein in shoot-root dry matter allocation in higher plants. Ann Bot 97:3–10
Beck EH (1994) The morphogenic response of plants to soil nitrogen: Adaptive regulation of biomass distribution and nitrogen metabolism by phytohormones. In: Schulze E-D (ed) Flux control in biological systems. Academic, Orlando, pp 119–151
Beck EH (1996) Regulation of shoot/root ratio by cytokinins from roots in Urtica dioica: opinion. Plant Soil 185:3–12
Bernard SM, Habash DZ (2009) The importance of cytosolic glutamine synthetase in nitrogen assimilation and recycling. New Phytol 182:608–620
Bloom AJ, Chapin FS, Mooney HA (1985) Resource limitation in plants—an economic analogy. Annu Rev Ecol Syst 16:363–392
Britto DT, Kronzucker HJ (2005) Nitrogen acquisition, PEP carboxylase, and cellular pH homeostasis: new views on old paradigms. Plant Cell Environ 28:1396–1409
Brouwer R (1962) Nutritive influences on distribution of dry matter in the plant. Neth J Agric Sci 10:399–408
Brouwer R (1983) Functional equilibrium: sense or non-sense? Neth J Agric Sci 31:335–348
Buysse J, Smolders E, Merckx R (1993) The role of free sugars and amino acids in the regulation of biomass partitioning and plant growth. Plant Soil 155/156:191–194
Cheeseman JM, Barreiro R, Lexa M (1996) Plant growth modelling and the integration of shoot and root activities without communicating messengers: opinion. Plant Soil 185:51–64
Chen ZH, Jenkins G, Nimmo HG (2008) pH and carbon supply control the expression of phosphoenolpyruvate carboxylase kinase genes. Plant Cell Environ 31:1844–1850
Cheng C-L, Acedo GN, Cristinsin M, Conkling MA (1992) Sucose mimics the light induction of Arabidopsis nitrate reductase gene transcription. Proc Nat Acad Sci USA 89:1861–1864
Correa-Aragunde I, Graziano M, Lamattina L (2006) Nitric oxide plays a central role in determining lateral root development in tomato. Planta 218:900–905
Crawford NM (1995) Nitrate: nutrient and signal for plant growth. Plant Cell 7:859–868
De Kroon H, Visser EJW, Huber H, Mommer L, Hutchings M (2009) A modular concept of plant foraging behaviour: the interplay between local responses and systemic control. Plant Cell Environ 32:704–712
Deng M-D, Moureaux T, Cherel I, Boutin J-P, Caboche M (1991) Effects of nitrogen metabolites on the regulation and circadian expression of tobacco nitrate reductase. Plant Physiol Biochem 29:237–247
Du S, Zhang Y, Lin X, Wang Y, Tang C (2008) Regulation of nitrate reductase by nitric oxide in Chinese cabbage pakchoi (Brassica chinensis L.). Plant Cell Environ 31:195–204
Farrar J (1996) Regulation of root weight ratio is mediated by sucrose: opinion. Plant Soil 185:13–19
Fetene M, Beck E (1993) Reversal of sink-source relations in Urtica dioica L. plants by increasing cytokinin import into the shoot. Bot Acta 106:235–240
Forde B (2002) Local and long-range signaling pathways regulating plant responses to nitrate. Annu Rev Plant Biol 53:203–224
Forde B, Lorenzo H (2001) The nutritional control of root development. Plant Soil 232:51–68
Forde BG, Lea PJ (2007) Glutamate in plants: metabolism, regulation, and signalling. J Exp Bot 58:2339–2358
Forde BG, Walch-Liu P (2009) Nitrate and glutamate as environmental cues for behavioural responses in plant roots. Plant Cell Environ 32:682–693
Foyer CH, Parry M, Noctor G (2003) Markers and signals associated with nitrogen assimilation in higher plants. J Exp Bot 382:585–593
Geßler A, Kopriva S, Rennenberg H (2004) Regulation of nitrate uptake of trees at the whole plant level: interaction between nitrogen compounds, cytokinins and carbon metabolism. Tree Physiol 24:1313–1321
Gifford ML, Dean A, Gutierrez RA, Corruzi G, Birnbaum KD (2008) Cell-specific nitrogen responses mediate developmental plasticity. Proc Nat Acad Sci USA 105:803–808
Hänsch R, Fessel DG, Hoffmann C, Hesberg C, Hoffmann G, Walch-Liu P, Engels C, Kruse J, Rennenberg H, Kaiser WM, Mendel R-R (2001) Tobacco plants that lack expression of a functional nitrate reductase in the roots show changes in growth rates and metabolite accumulation. J Exp Bot 52:1251–1258
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass allocation. Trends Plant Sci 12:610–617
Hodge A, Berta G, Doussan C, Merchan F, Crespi M (2009) Plant root growth, architecture and function. Plant Soil 321:153–187
Hodges M (2002) Enzyme redundancy and the importance of 2-oxoglutarate in plant ammonium assimilation. J Exp Bot 53:905–916
Imsande J, Touraine B (1994) N demand and the regulation of nitrate uptake. Plant Physiol 105:3–7
Jackson MB (1993) Are plant hormones involved in root to shoot communication? Adv Bot Res 19:104–187
Kreuzwieser J, Heschbach C, Stulen I, Wirsema P, Vaalburg W, Rennenberg H (1997) Interactions of NH +4 and L-glutamate with NO −3 transport processes of non-mycorrhizal Fagus sylvatica roots. J Exp Bot 312:1431–1438
Kruse J, Adams MA (2008) Integrating two physiological approaches helps relate plant respiration to growth of Pinus radiata. New Phytol 180:841–852
Kruse J, Hetzger I, Hänsch R, Mendel R-R, Walch-Liu P, Engels C, Rennenberg H (2002) Elevated pCO2 favours nitrate reduction in the roots of wild-type tobacco (Nicotiana tabacum cv. Gat.) and significantly alters N-metabolism in transformants lacking functional nitrate reductase in the roots. J Exp Bot 379:2351–2367
Kruse J, Hetzger I, Mai C, Polle A, Rennenberg H (2003a) Elevated pCO2 affects N-metabolism of young poplar plants (Populus tremula × P. alba) differently at deficient and sufficient N-supply. New Phytol 157:65–81
Kruse J, Hetzger I, Hänsch R, Mendel R-R, Rennenberg H (2003b) Evaluation of the effects of elevated pCO2 on C- and N-metabolism in wild type and transgenic tobacco exhibiting an altered C/N-balance by metabolite analysis. Plant Biol 5:540–549
Kruse J, Kopriva S, Hänsch R, Mendel R-R, Rennenberg H (2007) Interaction of sulphur and nitrogen nutrition in tobacco (Nicotiana tabacum) plants: significance of nitrogen source and root nitrate reductase. Plant Biol 9:638–646
Lam H-M, Chiu J, Hsieh M-H, Meisel L, Oliveira IC, Shin M, Corruzi G (1998) Glutamate receptor genes in plants. Nature 396:125–126
Lambers H (1983) The ‘functional equilibrium’: nibbling on the edges of a paradigm. Neth J Agric Sci 31:305–311
Lea PJ, Azevedo RA (2006) Nitrogen use efficiency. 1. Uptake of nitrogen from the soil. Ann Appl Biol 149:243–247
Lea PJ, Azevedo RA (2007) Nitrogen use efficiency. 2. Amino acid metabolism. Ann Appl Biol 151:269–275
Lea UM, ten Hoopen F, Provan F, Kaiser WM, Meyer C, Lillo C (2004) Mutation of the regulatory phosphorylation site of tobacco nitrate reductase results in high nitrite excretion and NO emission from leaf and root tissue. Planta 219:59–65
Lejay L, Gansel X, Cerezo M, Tillard P, Muller C, Krapp A, von Wiren N, Daniel-Vedele F, Gojon A (2003) Regulation of root ion transporters by photosynthesis: functional importance and relation with hexokinase. Plant Cell 15:2218–2232
Lexa M, Cheeseman JM (1997) Growth and nitrogen relations in reciprocal grafts of wild-type and nitrate reductase-deficient mutants of pea (Pisum sativum L. var. Juneau). J Exp Bot 311:1241–1250
Malamy J, Ryan K (2001) Environmental regulation of lateral root initiation in Arabidopsis. Plant Physiol 127:899–909
Marschner H (1995) Mineral nutrition of higher plants, vol 2. Academic; Harcourt Brace & Company, London
Miller AJ, Cramer MD (2004) Root nitrogen acquisition and assimilation. Plant Soil 274:1–36
Miller AJ, Fan X, Orsel M, Smith SJ, Wells DM (2007) Nitrate transport and signalling. J Exp Bot 58:2297–2306
Müller AJ (1983) Genetic analysis of nitrate reductase-deficient tobacco plants regenerated from mutant cells: evidence for duplicate structural genes. Mol Gen Genet 192:275–281
Nagel KA, Schurr U, Walter A (2006) Dynamics of root growth stimulation in Nicotiana tabacum in increasing light intensity. Plant Cell Environ 29:1936–1945
Neill SJ, Desikan R, Hancock JT (2003) Nitric oxide signalling in plants. New Phytol 159:11–35
Pagnussat GC, Simontacchi M, Puntarulo S, Lamattina L (2002) Nitric oxide is required for root organogenesis. Plant Physiol 129:954–956
Peuke AD (2000) Xylem and phloem transport, asimilation and partitioning of nitrogen in Ricinus communis under several nutritional conditions. In: MA Martins-Loucao, SH Lips (eds) Nitrogen in a sustainable ecosystem: from the cell to the plant, pp 211–225
Plaxton WC, Podesta FE (2006) The functional organization and control of plant respiration. Crit Rev Plant Sci 25:159–198
Raab TK, Terry N (1994) Nitrogen-source regulation of growth and photosynthesis in Beta vulgaris L. Plant Physiol 105:1159–1166
Rahayu YS, Walch-Liu P, Neumann G, von Wirén N, Bangerth F (2005) Root-derived cytokinins as long-distance signals for NO −3 -induced stimulation of leaf growth. J Exp Bot 414:1143–1152
Remans T, Pervent M, Filleur S, Diatloff E, Mounier E, Tillard P, Forde BG, Gojon A (2006) The Arabidopsis NRT1.1 transporter participates in the signalling pathway triggering root colonization of nitrate-rich patches. Proc Nat Acad Sci USA 103:19206–19211
Reynolds JF, Chen J (1996) Modelling whole plant allocation in relation to carbon and nitrogen supply: coordination versus optimization: opinion. Plant Soil 185:65–74
Scheible W-R, Lauerer M, Schulze E-D, Caboche M, Stitt M (1997a) Accumulation of nitrate in the shoot acts as a signal to regulate shoot-root allocation in tobacco. Plant J 11:671–691
Scheible W-R, Gonzalez-Fontes A, Lauerer M, Müller-Röber B, Caboche M, Stitt M (1997b) Nitrate acts as a signal to induce organic acid metabolism and repress starch metabolism in tobacco. Plant Cell 9:783–798
Scheurwater I, Koren M, Lambers H, Atkin OK (2002) The contribution of roots and shoots to whole plant nitrate reduction in fast- and slow-growing grass species. J Exp Bot 374:1635–1642
Schortemeyer M, Feil B (1996) Root morphology of maize under homogenous or spatially separated supply of ammonium and nitrate at three concentration ratios. J Plant Nutr 19:1089–1097
Stitt M, Scheible W-R (1998) Understanding allocation to shoot and root growth will require molecular information about which compounds act as signals for the plant nutrient status, and how meristem activity and cellular growth are regulated: Opinion. Plant Soil 201:259–263
Stitt M, Müller C, Matt P, Gibon Y, Carillo P, Morcuende R, Scheible W-R, Krapp A (2002) Steps towards an integrated view of nitrogen metabolism. J Exp Bot 53:959–970
Stoimenova M, Hänsch R, Mendel R, Gimmler H, Kaiser WM (2003a) The role of nitrate reduction in the anoxic metabolism of roots I. Characterization of root morphology and normoxic metabolism of wild type tobacco and a transformant lacking root nitrate reductase. Plant Soil 253:145–153
Stoimenova M, Libourel IGL, Ratcliffe RG, Kaiser WM (2003b) The role of nitrate reduction in the anoxic metabolism of roots II. Anoxix metabolism of tobacco roots with or without nitrate reductase activity. Plant Soil 253:155–167
Takei K, Ueda N, Aoki K, Kuromori T, Hirayama T, Shinozaki K, Yamaya T, Sakakibara H (2004) AtIPT3 is a key determinant of nitrate-dependent cytokinin biosynthesis in Arabidopsis. Plant Cell Physiol 45:1053–1062
Touraine B, Clarkson DT and Muller B (1994) Regulation of nitrate uptake at the whole plant level. In: Roy J, Garnier E (eds) A whole plant perspective on carbon-nitrogen interactions, pp 11–30
Tschoep H, Gibon Y, Carillo P, Armengaud P, Szecowka M, Nunes-Nesi A, Fernie AR, Koehl K, Stitt M (2009) Adjustment of growth and central metabolism to a mild but sustained nitrogen-limitation in Arabidopsis. Plant Cell Environ 32:300–318
Van der Werf A, Nagel OW (1996) Carbon allocation to shoots and roots is mediated by cytokinins and sucrose. Plant Soil 185:21–32
Van der Werf A, Enserink T, Smit B, Booij R (1993) Allocation of carbon and nitrogen as a function of the internal nitrogen status of a plant: modelling allocation under non-steady-state situations. Plant Soil 155(156):183–186
Vidmar J, Zhuo D, Siddiqi M, Schjoerring J, Touraine B, Glass A (2000) Regulation of high-affinity nitrate influx by nitrogen pools in roots of barley. Plant Physiol 123:307–318
Walch-Liu P, Forde BG (2008) Nitrate signalling mediated by the NRT1.1 nitrate transporter antagonizes L-glutamate-induced changes in root architecture. Plant J 54:820–828
Walch-Liu P, Neumann G, Bangerth F, Engels C (2000) Rapid effects of nitrogen form on leaf morphogenesis in tobacco. J Exp Bot 343:227–237
Walch-Liu P, Ivanov II, Filleur S, Gan Y, Remans T, Forde BG (2006) Nitrogen regulation of root branching. Ann Bot 97:875–881
Warner RL, Kleinhofs A, Muehlbauer FJ (1982) Characterization of nitrate reductase-deficient mutants in pea. Crop Sci 22:389–393
Wilson JB (1988) A review of evidence on the control of shoot: root ratio, in relation to models. Ann Bot 61:433–449
Woolfolk WT, Friend AL (2003) Growth response of cottonwood roots to varied NH4: NO3 ratios in enriched patches. Tree Physiol 23:427–432
Yamasaki H (2005) The NO world for plants: achieving balance in an open system. Plant Cell Environ 28:78–84
Zhang H, Jennings A, Barlow PW, Forde BG (1999) Dual pathways for regulation of root branching by nitrate. Proc Nat Acad Sci USA 96:6529–6534
Zhang H, Rong H, Pilbeam D (2007) Signalling mechanisms underlying the morphological responses of the root system to nitrogen in Arabidopsis thaliana. J Exp Bot 58:2329–2338
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: A.C. Borstlap.
Rights and permissions
About this article
Cite this article
Kruse, J., Hänsch, R., Mendel, R.R. et al. The role of root nitrate reduction in the systemic control of biomass partitioning between leaves and roots in accordance to the C/N-status of tobacco plants. Plant Soil 332, 387–403 (2010). https://doi.org/10.1007/s11104-010-0305-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-010-0305-6