Abstract
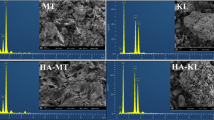
The sorption of three organic contaminants with different structure and polarity including non-polar phenanthrene (PHEN), 1,2,4,5-tetrachlorobenzene (TeCB), and polar 1,2-dichlorobenzene (DCB) onto original kaolinite, smectite, vermiculite, and fulvic acid (FA)/humic acid (HA)–clay complexes were investigated, and possible sorption mechanisms were inferred from sorption isotherms and characteristics of humic substances (HS) and HS–mineral complexes. Results showed smectite and vermiculite had stronger sorption ability than kaolinite, and the adsorbed amount of DCB was much higher than that of PHEN and TeCB on each clay. Due to FA/HA-facilitated hydrophobic interaction, FA/HA–clay complexes except FA–vermiculite complex showed a stronger affinity for PHEN and TeCB than the original clays, particularly for HA–clay complexes. The non-linearity parameter values of n for all the Freundlich sorption isotherms of DCB were greater than 1, indicating that clays possessed some unique sites with strong affinity and capacity to sorb DCB from aqueous solutions. FA/HA did not significantly affect the sorption of polar DCB on clays, implying sorption of DCB on clays was probably due to polar interactions between the polar group of DCB and clays. Cation-π bonding between PHEN and iron cation was directly evidenced by X-ray photoelectron spectroscopy, and FA impeded the sorption of PHEN on vermiculite by occupation of iron cation sites. This study will benefit understanding behaviors of contaminants in the soil environments.






Similar content being viewed by others
References
Brunetti, G., Mezzapesa, G. N., Traversa, A., Bonifacio, E., Farrag, K., Senesi, N., & D’Orazio, V. (2016). Characterization of clay- and silt-sized fractions and corresponding humic acids along a terra rossa soil profile. Clean-Soil Air Water, 44(10), 1375–1384.
Celis, R., Hermosin, M. C., Cox, L., & Cornejo, J. (1999). Sorption of 2,4-dichlorophenoxyacetic acid by model particles simulating naturally occurring soil colloids. Environmental Science and Technology, 33, 1200–1206.
Celis, R., Jonge, H. D., Jonge, L. W. D., Real, M., Hermosin, M. C., & Cornejo, J. (2006). The role of mineral and organic components in phenanthrene and dibenzofuran sorption by soil. European Journal of Soil Science, 57(3), 308–319.
Du, H. H., Chen, W. L., Cai, P., Rong, X. M., Dai, K., Peacock, C. L., & Huang, Q. Y. (2016). Cd(II) sorption on montmorillonite-humic acid-bacteria composites. Scientific Reports, 6, 19499.
Han, L. F., Sun, K., Jin, J., & Xing, B. S. (2016). Some concepts of soil organic carbon characteristics and mineral interaction from a review of literature. Soil Biology and Biochemistry, 94, 107–121.
Hengpraprom, S., Lee, C. M., & Coates, J. T. (2006). Sorption of humic acids and α-endosulfan by clay minerals. Environmental Toxicology and Chemistry, 25(1), 11–17.
Hundal, L. S., Thompson, M. L., Laird, D. A., & Carmo, A. M. (2001). Sorption of phenanthrene by reference smectites. Environmental Science and Technology, 35(17), 3456–3461.
Karickhoff, S. W., Brown, D. S., & Scott, T. A. (1979). Sorption of hydrophobic pollutants on natural sediments. Water Research, 13(3), 241–248.
Liu, W. P., Gan, J. Y., Papiernik, S. K., & Yates, S. R. (2000). Structural influences in relative sorptivity of chloroacetanilide herbicides on soil. Journal of Agricultural and Food Chemistry, 48(9), 4320–4325.
Means, J. C., Wood, S. G., Hassett, J. J., & Banwart, W. L. (1980). Sorption of polynuclear aromatic hydrocarbons by sediments and soils. Environmental Science and Technology, 14(12), 1524–1528.
Namjesnik-Dejanovic, K., Maurice, P. A., Aiken, G. R., Cabaniss, S., Chin, Y. P., & Pullin, M. J. (2000). Adsorption and fractionation of a muck fulvic acid on kaolinite and goethite at pH 3.7, 6, and 8. Soil Science, 165(7), 545–559.
Pinnavaia, T. J., Hall, P. L., Cady, S. S., & Mortland, M. M. (1974). Aromatic radical cation formation on the intracrystal surfaces of transition metal layer lattice silicates. Journal of Physical Chemistry, 78(10), 994–999.
Rastegari, M., Saeedi, M., Mollahosseini, A., & Ayatynia, M. (2016). Phenanthrene sorption onto kaolinite; heavy metals and organic matter effects. International Journal of Environmental Research, 10(3), 441–448.
Simoes, A., Stringfellow, A. M., Smallman, D., Beaven, R., Marshall, J., Powrie, W., & Potter, H. A. B. (2011). Sorption of organic contaminants by Oxford clay and Mercia mudstone landfill liners. Quarterly Journal of Engineering Geology and Hydrogeology, 44(3), 345–360.
Smernik, R. J., & Kookana, R. S. (2015). The effects of organic matter–mineral interactions and organic matter chemistry on diuron sorption across a diverse range of soils. Chemosphere, 119, 99–104.
Specht, C. H., Kumke, M. U., & Frimmel, F. H. (2000). Characterization of NOM adsorption to clay minerals by size exclusion chromatography. Water Research, 34(16), 4063–4069.
Wang, M. C., & Huang, P. M. (1989). Pyrogallol transformations as catalyzed by nontronite, bentonite, and kaolinite. Clays and Clay Minerals, 37(6), 525–531.
Wang, K. J., & Xing, B. S. (2005). Structural and sorption characteristics of adsorbed humic acid on clay minerals. Journal of Environmental Quality, 34(1), 342–349.
Werner, D., Garratt, J. A., & Pigott, G. (2013). Sorption of 2,4-D and other phenoxy herbicides to soil, organic matter, and minerals. Journal of Soils and Sediments, 13(1), 129–139.
Wu, P. X., Tang, Y. N., Wang, W. M., Zhu, N. W., Li, P., Wu, J. H., Dang, Z., & Wang, X. D. (2011). Effect of dissolved organic matter from Guangzhou landfill leachate on sorption of phenanthrene by montmorillonite. Journal of Colloid and Interface Science, 361(2), 618–627.
Zalba, P., Amiotti, N. M., Galantini, J. A., & Pistola, S. (2016). Soil humic and fulvic acids from different land-use systems evaluated by E4/E6 ratios. Communications in Soil Science and Plant Analysis, 47(13–14), 1675–1679.
Zhang, L. C., Luo, L., & Zhang, S. Z. (2012). Integrated investigations on the adsorption mechanisms of fulvic and humic acids on three clay minerals. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 406(14), 84–90.
Zhang, Y. J., Zhu, D. Q., & Yu, H. X. (2008). Sorption of aromatic compounds to clay mineral and model humic substance–clay complex: effects of solute structure and exchangeable cation. Journal of Environmental Quality, 37, 817–823.
Acknowledgements
This work was supported by the National Natural Science Foundation of China [grant numbers 21407053, 41371444, and 31300422]; the Natural Science Foundation from Educational Commission of Anhui Province, China [grant number KJ2016A636]; and the Natural Science Foundation of Anhui Province, China [grant numbers 1308085MB28, 1408085QC47].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 302 kb)
Rights and permissions
About this article
Cite this article
Zhang, L., Xiang, P., Bao, X. et al. The Influence of Humic Substances on the Sorption of Three Organic Contaminants with Different Structure and Polarity to Clay Minerals. Water Air Soil Pollut 228, 199 (2017). https://doi.org/10.1007/s11270-017-3380-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3380-y