Abstract
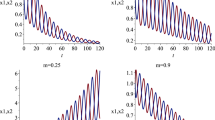
Hybrid models for gene expression combine stochastic and deterministic representations of the underlying biophysical mechanisms. According to one of the simplest hybrid formalisms, protein molecules are produced in randomly occurring bursts of a randomly distributed size while they are degraded deterministically. Here, we use this particular formalism to study two key regulatory motifs—the autoregulation loop and the toggle switch. The distribution of burst times is determined and used as a basis for the development of exact simulation algorithms for gene expression dynamics. For the autoregulation loop, the simulations are compared to an analytic solution of a master equation. Simulations of the toggle switch reveal a number of qualitatively distinct scenarios with implications for the modelling of cell-fate selection.






Similar content being viewed by others
Notes
Alternatively, n>0 could be required in place of a 1>0, with a 1<0 corresponding to positive feedback.
References
Alon, U. (2007). An introduction to systems biology: design principles of biological circuits. London: Chapman & Hall/CRC.
Andrecut, M., & Kauffman, S. A. (2006). Noise in genetic toggle switch models. J. Int. Bioinform, 23.
Andrecut, M., Halley, J. D., Winkler, D. A., & Huang, S. (2011). A general model for binary cell fate decision gene circuits with degeneracy: indeterminacy and switch behavior in the absence of cooperativity. PLoS ONE, 6, e19358.
Bicout, D. J. (1997). Green’s functions and first passage time distributions for dynamic instability of microtubules. Phys. Rev. E, 56(6), 6656–6667.
Bobrowski, A., Lipniacki, T., Pichór, K., & Rudnicki, R. (2007). Asymptotic behavior of distributions of mrna and protein levels in a model of stochastic gene expression. J. Math. Anal. Appl., 333, 753–769.
Bokes, P. (2010). Genetic regulatory networks. PhD thesis, University of Nottingham.
Bokes, P., King, J. R., & Loose, M. (2009). A bistable genetic switch which does not require high co-operativity at the promoter: a two-timescale model for the PU. 1–GATA-1 interaction. Math. Med. Biol., 26, 117–132.
Bokes, P., King, J. R., Wood, A. T. A., & Loose, M. (2012). Multiscale stochastic modelling of gene expression. J. Math. Biol.
Chang, H. H., Hemberg, M., Barahona, M., Ingber, D. E., & Huang, S. (2008). Transcriptome-wide noise controls lineage choice in mammalian progenitor cells. Nature, 453, 544–547.
Cherry, J. L., & Adler, F. R. (2000). How to make a biological switch. J. Theor. Biol., 203, 117–133.
Chow, C. C., & White, J. A. (1996). Spontaneous action potentials due to channel fluctuations. Biophys. J., 71, 3013–3021.
Cinquemani, E., Porreca, R., Ferrari-Trecate, G., & Lygeros, J. (2008). Subtilin production by bacillus subtilis: stochastic hybrid models and parameter identification. IEEE Trans. Autom. Control, 53, 38–50.
Cox, D. R., & Oakes, D. (1984). Analysis of survival data. Boca Raton: Chapman & Hall/CRC.
Crudu, A., Debussche, A., & Radulescu, O. (2009). Hybrid stochastic simplifications for multiscale gene networks. BMC Syst. Biol., 3, 89.
Davis, M. H. A. (1984). Piecewise-deterministic Markov processes: a general class of non-diffusion stochastic models. J. R. Stat. Soc. B, 46, 353–388.
De Jong, H. (2002). Modeling and simulation of genetic regulatory systems: a literature review. J. Comput. Biol., 9, 67–103.
Friedman, N., Cai, L., & Xie, X. S. (2006). Linking stochastic dynamics to population distribution: an analytical framework of gene expression. Phys. Rev. Lett., 97, 168302.
Gardner, T. S., Cantor, C. R., & Collins, J. J. (2000). Construction of a genetic toggle switch in Escherichia coli. Nature, 403, 339–342.
Gillespie, D. T. (1976). A general method for numerically simulating stochastic time evolution of coupled chemical reactions. J. Comput. Phys., 22, 403–434.
Gillespie, D. T. (1977). Exact stochastic simulation of coupled chemical reactions. J. Phys. Chem., 81, 2340–2361.
Griffith, J. S. (1968a). Mathematics of cellular control processes. I. Negative feedback to one gene. J. Theor. Biol., 20, 202–208.
Griffith, J. S. (1968b). Mathematics of cellular control processes. II. Positive feedback to one gene. J. Theor. Biol., 20, 209–216.
Higham, D. J., Intep, S., Mao, X., & Szpruch, L. (2011). Hybrid simulation of autoregulation within transcription and translation. BIT Numer. Math., 51, 177–196.
Hu, M., Krause, D., Greaves, M., Sharkis, S., Dexter, M., Heyworth, C., & Enver, T. (1997). Multilineage gene expression precedes commitment in the hemopoietic system. Genes Dev., 11, 774–785.
Huang, S., Guo, Y. P., May, G., & Enver, T. (2007). Bifurcation dynamics in lineage-commitment in bipotent progenitor cells. Dev. Biol., 305, 695–713.
Keener, J. P., & Newby, J. M. (2011). Perturbation analysis of spontaneous action potential initiation by stochastic ion channels. Phys. Rev. E, 84, 011918.
Keener, J., & Sneyd, J. (2008). Mathematical physiology: cellular physiology. Berlin: Springer.
Kepler, T. B., & Elston, T. C. (2001). Stochasticity in transcriptional regulation: origins, consequences, and mathematical representations. Biophys. J., 81, 3116–3136.
Laslo, P., Spooner, C. J., Warmflash, A., Lancki, D. W., Lee, H. J., Sciammas, R., Gantner, B. N., Dinner, A. R., & Singh, H. (2006). Multilineage transcriptional priming and determination of alternate hematopoietic cell fates. Cell, 126, 755–766.
Lei, J., Mackey, M. C., Yvinec, R., & Zhuge, C. (2012). Adiabatic reduction of a piecewise deterministic Markov model of stochastic gene expression with bursting transcription. arXiv:1202.5411.
Lipniacki, T., Paszek, P., Marciniak-Czochra, A., Brasier, A. R., & Kimmel, M. (2006). Transcriptional stochasticity in gene expression. J. Theor. Biol., 238, 348–367.
Loinger, A., Lipshtat, A., Balaban, N. Q., & Biham, O. (2007). Stochastic simulations of genetic switch systems. Phys. Rev. E, 75, 021904.
Lu, P., Vogel, C., Wang, R., Yao, X., & Marcotte, E. M. (2007). Absolute protein expression profiling estimates the relative contributions of transcriptional and translational regulation. Nat. Biotechnol., 25, 117–124.
Mackey, M. C., & Tyran-Kaminska, M. (2008). Dynamics and density evolution in piecewise deterministic growth processes. Ann. Pol. Math., 94, 111–129.
Mackey, M. C., Tyran-Kaminska, M., & Yvinec, R. (2011). Molecular distributions in gene regulatory dynamics. J. Theor. Biol., 274, 84–96.
Munsky, B., & Khammash, M. (2010). Identification from stochastic cell-to-cell variation: a genetic switch case study. IET Syst. Biol., 4, 356–366.
Murray, J. D. (2003). Mathematical biology. Berlin: Springer.
Newby, J. M. (2011). Isolating intrinsic noise sources in a stochastic genetic switch. arXiv:1111.1415.
Newby, J., & Bressloff, P. C. (2010). Local synaptic signaling enhances the stochastic transport of motor-driven cargo in neurons. Phys. Biol., 7, 036004.
Novák, B., & Tyson, J. J. (2008). Design principles of biochemical oscillators. Nat. Rev. Mol. Cell Biol., 9(12), 981–991.
Othmer, H. G., Dunbar, S. R., & Alt, W. (1988). Models of dispersal in biological systems. J. Math. Biol., 26(3), 263–298.
Raj, A., Peskin, C. S., Tranchina, D., Vargas, D. Y., & Tyagi, S. (2006). Stochastic mRNA synthesis in mammalian cells. PLoS Biol., 4, e309.
Robert, C. P., & Casella, G. (2004). Monte Carlo statistical methods. Berlin: Springer.
Shea, M. A., & Ackers, G. K. (1985). The OR control system of bacteriophage lambda. A physical-chemical model for gene regulation. J. Mol. Biol., 181, 211–230.
Singh, A., & Hespanha, J. P. (2010). Stochastic hybrid systems for studying biochemical processes. Philos. Trans. R. Soc. Lond. A, 368, 4995–5011.
Swiers, G., Patient, R., & Loose, M. (2006). Genetic regulatory networks programming hematopoietic stem cells and erythroid lineage specification. Dev. Biol., 294, 525–540.
Taniguchi, Y., Choi, P. J., Li, G. W., Chen, H., Babu, M., Hearn, J., Emili, A., & Xie, X. S. (2010). Quantifying E. coli proteome and transcriptome with single-molecule sensitivity in single cells. Science, 329, 533–538.
Tian, T., & Burrage, K. (2006). Stochastic models for regulatory networks of the genetic toggle switch. Proc. Natl. Acad. Sci. USA, 103, 8372–8377.
Tyson, J. J., Chen, K. C., & Novak, B. (2003). Sniffers, buzzers, toggles and blinkers: dynamics of regulatory and signaling pathways in the cell. Curr. Opin. Cell Biol., 15, 221–231.
van Kampen, N. G. (2006). Stochastic processes in physics and chemistry. Amsterdam: Elsevier.
Zeiser, S., Franz, U., Wittich, O., & Liebscher, V. (2008). Simulation of genetic networks modelled by piecewise deterministic Markov processes. IET Syst. Biol., 2, 113–135.
Zeiser, S., Franz, U., Müller, J., & Liebscher, V. (2009). Hybrid modeling of noise reduction by a negatively autoregulated system. Bull. Math. Biol., 71, 1006–1024.
Zeiser, S., Franz, U., & Liebscher, V. (2010). Autocatalytic genetic networks modeled by piecewise-deterministic Markov processes. J. Math. Biol., 60, 207–246.
Zenklusen, D., Larson, D. R., & Singer, R. H. (2008). Single-rna counting reveals alternative modes of gene expression in yeast. Nat. Struct. Mol. Biol., 15, 1263–1271.
Acknowledgements
P. Bokes was supported by the European Commission under Marie Curie Early Stage Researcher Training (contract no. MEST-CT-2005-020723) and also by the Slovak Research and Development Agency (contract no. APVV-0134-10). J. King gratefully acknowledges the funding of the BBSRC/EPSRC (reference no. BB/D008522/1) and of the Royal Society and Wolfson Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bokes, P., King, J.R., Wood, A.T.A. et al. Transcriptional Bursting Diversifies the Behaviour of a Toggle Switch: Hybrid Simulation of Stochastic Gene Expression. Bull Math Biol 75, 351–371 (2013). https://doi.org/10.1007/s11538-013-9811-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-013-9811-z