Abstract
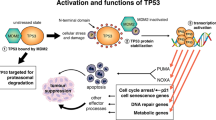
The tumour suppressor p53 is a central player in cellular DNA damage responses. P53 is upregulated and activated by genotoxic stress and induces a transcriptional programme with effectors promoting apoptosis, cell cycle arrest, senescence and DNA repair. For the best part of the last three decades, these DNA damage-related programmes triggered by p53 were unequivocally regarded as the major if not sole mechanism by which p53 exerts its tumour suppressor function. However, this interpretation has been challenged by a number of recent in vivo studies, demonstrating that mice which are defective in inducing p53-dependent apoptosis, cell cycle arrest and senescence suppress thymic lymphoma as well as wild-type p53 expressing animals. Consequently, the importance of DNA damage responses for p53-mediated tumour suppression has been questioned. In this review, I summarize current knowledge on p53-controlled DNA damage responses and argue that these activities, while their role has certainly changed, remain an important feature of p53 biology with relevance for cancer therapy and tumour suppression.


Similar content being viewed by others
References
Adimoolam S, Ford JM (2002) p53 and DNA damage-inducible expression of the xeroderma pigmentosum group C gene. Proc Natl Acad Sci USA 99(20):12985–12990. doi:10.1073/pnas.202485699
Adnane J, Jackson RJ, Nicosia SV, Cantor AB, Pledger WJ, Sebti SM (2000) Loss of p21WAF1/CIP1 accelerates Ras oncogenesis in a transgenic/knockout mammary cancer model. Oncogene 19(47):5338–5347. doi:10.1038/sj.onc.1203956
Agarwal ML, Agarwal A, Taylor WR, Stark GR (1995) p53 controls both the G2/M and the G1 cell cycle checkpoints and mediates reversible growth arrest in human fibroblasts. Proc Natl Acad Sci USA 92(18):8493–8497
Akyuz N, Boehden GS, Susse S et al (2002) DNA substrate dependence of p53-mediated regulation of double-strand break repair. Mol Cell Biol 22(17):6306–6317
Allen MA, Andrysik Z, Dengler VL et al (2014) Global analysis of p53-regulated transcription identifies its direct targets and unexpected regulatory mechanisms. eLife 3:e02200. doi:10.7554/eLife.02200
Attardi LD, Reczek EE, Cosmas C et al (2000) PERP, an apoptosis-associated target of p53, is a novel member of the PMP-22/gas3 family. Genes Dev 14(6):704–718
Aziz MH, Shen H, Maki CG (2011) Acquisition of p53 mutations in response to the non-genotoxic p53 activator Nutlin-3. Oncogene 30(46):4678–4686. doi:10.1038/onc.2011.185
Azzam EI, de Toledo SM, Pykett MJ, Nagasawa H, Little JB (1997) CDC2 is down-regulated by ionizing radiation in a p53-dependent manner. Cell Growth Differ Mol Biol J Am Assoc Cancer Res 8(11):1161–1169
Barboza JA, Liu G, Ju Z, El-Naggar AK, Lozano G (2006) p21 delays tumor onset by preservation of chromosomal stability. Proc Natl Acad Sci USA 103(52):19842–19847. doi:10.1073/pnas.0606343104
Bargonetti J, Friedman PN, Kern SE, Vogelstein B, Prives C (1991) Wild-type but not mutant p53 immunopurified proteins bind to sequences adjacent to the SV40 origin of replication. Cell 65(6):1083–1091
Berkers CR, Maddocks OD, Cheung EC, Mor I, Vousden KH (2013) Metabolic regulation by p53 family members. Cell Metab 18(5):617–633. doi:10.1016/j.cmet.2013.06.019
Berns EM, Foekens JA, Vossen R et al (2000) Complete sequencing of TP53 predicts poor response to systemic therapy of advanced breast cancer. Cancer Res 60(8):2155–2162
Bertheau P, Turpin E, Rickman DS et al (2007) Exquisite sensitivity of TP53 mutant and basal breast cancers to a dose-dense epirubicin-cyclophosphamide regimen. PLoS Med 4(3):e90. doi:10.1371/journal.pmed.0040090
Bieging KT, Attardi LD (2012) Deconstructing p53 transcriptional networks in tumor suppression. Trends Cell Biol 22(2):97–106. doi:10.1016/j.tcb.2011.10.006
Bieging KT, Mello SS, Attardi LD (2014) Unravelling mechanisms of p53-mediated tumour suppression. Natl Rev Cancer 14(5):359–370. doi:10.1038/nrc3711
Bill CA, Yu Y, Miselis NR, Little JB, Nickoloff JA (1997) A role for p53 in DNA end rejoining by human cell extracts. Mutat Res 385(1):21–29
Bommer GT, Gerin I, Feng Y et al (2007) p53-Mediated activation of miRNA34 candidate tumor-suppressor genes. Curr Biol CB 17(15):1298–1307. doi:10.1016/j.cub.2007.06.068
Brady CA, Jiang D, Mello SS et al (2011) Distinct p53 transcriptional programs dictate acute DNA-damage responses and tumor suppression. Cell 145(4):571–583. doi:10.1016/j.cell.2011.03.035
Braun CJ, Zhang X, Savelyeva I et al (2008) p53-Responsive micrornas 192 and 215 are capable of inducing cell cycle arrest. Cancer Res 68(24):10094–10104. doi:10.1158/0008-5472.can-08-1569
Bristow RG, Hu Q, Jang A et al (1998) Radioresistant MTp53-expressing rat embryo cell transformants exhibit increased DNA-dsb rejoining during exposure to ionizing radiation. Oncogene 16(14):1789–1802. doi:10.1038/sj.onc.1201935
Brosh R, Rotter V (2009) When mutants gain new powers: news from the mutant p53 field. Nat Rev Cancer 9(10):701–713
Brown CJ, Lain S, Verma CS, Fersht AR, Lane DP (2009) Awakening guardian angels: drugging the p53 pathway. Natl Rev Cancer 9(12):862–873. doi:10.1038/nrc2763
Brugarolas J, Chandrasekaran C, Gordon JI, Beach D, Jacks T, Hannon GJ (1995) Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature 377(6549):552–557
Bunz F, Dutriaux A, Lengauer C et al (1998) Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 282(5393):1497–1501
Bunz F, Hwang PM, Torrance C et al (1999) Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J Clin Investig 104(3):263–269. doi:10.1172/jci6863
Carvajal LA, Manfredi JJ (2013) Another fork in the road—life or death decisions by the tumour suppressor p53. EMBO Rep 14(5):414–421. doi:10.1038/embor.2013.25
Chan TA, Hermeking H, Lengauer C, Kinzler KW, Vogelstein B (1999) 14-3-3Sigma is required to prevent mitotic catastrophe after DNA damage. Nature 401(6753):616–620. doi:10.1038/44188
Chang BD, Broude EV, Dokmanovic M et al (1999a) A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res 59(15):3761–3767
Chang BD, Xuan Y, Broude EV et al (1999b) Role of p53 and p21waf1/cip1 in senescence-like terminal proliferation arrest induced in human tumor cells by chemotherapeutic drugs. Oncogene 18(34):4808–4818. doi:10.1038/sj.onc.1203078
Chang TC, Wentzel EA, Kent OA et al (2007) Transactivation of miR-34a by p53 broadly influences gene expression and promotes apoptosis. Mol Cell 26(5):745–752. doi:10.1016/j.molcel.2007.05.010
Chehab NH, Malikzay A, Stavridi ES, Halazonetis TD (1999) Phosphorylation of Ser-20 mediates stabilization of human p53 in response to DNA damage. Proc Natl Acad Sci USA 96(24):13777–13782
Chen J, Sadowski I (2005) Identification of the mismatch repair genes PMS2 and MLH1 as p53 target genes by using serial analysis of binding elements. Proc Natl Acad Sci USA 102(13):4813–4818. doi:10.1073/pnas.0407069102
Chen X, Ko LJ, Jayaraman L, Prives C (1996) p53 Levels, functional domains, and DNA damage determine the extent of the apoptotic response of tumor cells. Genes Dev 10(19):2438–2451
Cheng Q, Chen L, Li Z, Lane WS, Chen J (2009) ATM activates p53 by regulating MDM2 oligomerization and E3 processivity. EMBO J 28(24):3857–3867. doi:10.1038/emboj.2009.294
Chin KV, Ueda K, Pastan I, Gottesman MM (1992) Modulation of activity of the promoter of the human MDR1 gene by Ras and p53. Science 255(5043):459–462
Chipuk JE, Kuwana T, Bouchier-Hayes L et al (2004) Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303(5660):1010–1014
Chipuk JE, Bouchier-Hayes L, Kuwana T, Newmeyer DD, Green DR (2005) PUMA couples the nuclear and cytoplasmic proapoptotic function of p53. Science 309(5741):1732–1735
Chrisanthar R, Knappskog S, Lokkevik E et al (2008) CHEK2 mutations affecting kinase activity together with mutations in TP53 indicate a functional pathway associated with resistance to epirubicin in primary breast cancer. PLoS ONE 3(8):e3062. doi:10.1371/journal.pone.0003062
Christophorou MA, Ringshausen I, Finch AJ, Swigart LB, Evan GI (2006) The pathological response to DNA damage does not contribute to p53-mediated tumour suppression. Nature 443(7108):214–217. doi:10.1038/nature05077
Cinatl J, Speidel D, Hardcastle I, Michaelis M (2014) Resistance acquisition to MDM2 inhibitors. Biochem Soc Trans 42(4):752–757. doi:10.1042/bst20140035
Clarke AR, Purdie CA, Harrison DJ et al (1993) Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362(6423):849–852. doi:10.1038/362849a0
Cole AM, Ridgway RA, Derkits SE et al (2010) p21 loss blocks senescence following Apc loss and provokes tumourigenesis in the renal but not the intestinal epithelium. EMBO Mol Med 2(11):472–486. doi:10.1002/emmm.201000101
Concepcion CP, Han YC, Mu P et al (2012) Intact p53-dependent responses in miR-34-deficient mice. PLoS Genet 8(7):e1002797. doi:10.1371/journal.pgen.1002797
da Costa NM, Hautefeuille A, Cros MP et al (2012) Transcriptional regulation of thymine DNA glycosylase (TDG) by the tumor suppressor protein p53. Cell Cycle 11(24):4570–4578. doi:10.4161/cc.22843
Dahm-Daphi J, Hubbe P, Horvath F et al (2005) Nonhomologous end-joining of site-specific but not of radiation-induced DNA double-strand breaks is reduced in the presence of wild-type p53. Oncogene 24(10):1663–1672. doi:10.1038/sj.onc.1208396
Das S, Raj L, Zhao B et al (2007) Hzf Determines cell survival upon genotoxic stress by modulating p53 transactivation. Cell 130(4):624–637. doi:10.1016/j.cell.2007.06.013
de Souza-Pinto NC, Harris CC, Bohr VA (2004) p53 Functions in the incorporation step in DNA base excision repair in mouse liver mitochondria. Oncogene 23(39):6559–6568. doi:10.1038/sj.onc.1207874
de Toledo SM, Azzam EI, Keng P, Laffrenier S, Little JB (1998) Regulation by ionizing radiation of CDC2, cyclin A, cyclin B, thymidine kinase, topoisomerase IIalpha, and RAD51 expression in normal human diploid fibroblasts is dependent on p53/p21Waf1. Cell growth Differ Mol Biol J Am Assoc Cancer Res 9(11):887–896
Deng C, Zhang P, Harper JW, Elledge SJ, Leder P (1995) Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 82(4):675–684
Di Leonardo A, Linke SP, Clarkin K, Wahl GM (1994) DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev 8(21):2540–2551
Donehower LA, Harvey M, Slagle BL et al (1992) Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356(6366):215–221. doi:10.1038/356215a0
Doumont G, Martoriati A, Beekman C et al (2005) G1 checkpoint failure and increased tumor susceptibility in mice lacking the novel p53 target Ptprv. EMBO J 24(17):3093–3103. doi:10.1038/sj.emboj.7600769
Dudenhoffer C, Rohaly G, Will K, Deppert W, Wiesmuller L (1998) Specific mismatch recognition in heteroduplex intermediates by p53 suggests a role in fidelity control of homologous recombination. Mol Cell Biol 18(9):5332–5342
Dulic V, Kaufmann WK, Wilson SJ et al (1994) p53-dependent inhibition of cyclin-dependent kinase activities in human fibroblasts during radiation-induced G1 arrest. Cell 76(6):1013–1023
Dutta A, Ruppert JM, Aster JC, Winchester E (1993) Inhibition of DNA replication factor RPA by p53. Nature 365(6441):79–82. doi:10.1038/365079a0
El-Deiry WS, Kern SE, Pietenpol JA, Kinzler KW, Vogelstein B (1992) Definition of a consensus binding site for p53. Nat Genet 1(1):45–49. doi:10.1038/ng0492-45
El-Deiry WS, Tokino T, Velculescu VE et al (1993) WAF1, a potential mediator of p53 tumor suppression. Cell 75(4):817–825
El-Deiry WS, Harper JW, O’Connor PM et al (1994) WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 54(5):1169–1174
Erster S, Mihara M, Kim RH, Petrenko O, Moll UM (2004) In vivo mitochondrial p53 translocation triggers a rapid first wave of cell death in response to DNA damage that can precede p53 target gene activation. Mol Cell Biol 24(15):6728–6741
Espinosa JM, Verdun RE, Emerson BM (2003) p53 Functions through stress- and promoter-specific recruitment of transcription initiation components before and after DNA damage. Mol Cell 12(4):1015–1027
Fan S, El-Deiry WS, Bae I et al (1994) p53 Gene mutations are associated with decreased sensitivity of human lymphoma cells to DNA damaging agents. Cancer Res 54(22):5824–5830
Ford JM, Hanawalt PC (1995) Li-Fraumeni syndrome fibroblasts homozygous for p53 mutations are deficient in global DNA repair but exhibit normal transcription-coupled repair and enhanced UV resistance. Proc Natl Acad Sci USA 92(19):8876–8880
Ford JM, Hanawalt PC (1997) Expression of wild-type p53 is required for efficient global genomic nucleotide excision repair in UV-irradiated human fibroblasts. J Biol Chem 272(44):28073–28080
Fritsche M, Haessler C, Brandner G (1993) Induction of nuclear accumulation of the tumor-suppressor protein p53 by DNA-damaging agents. Oncogene 8(2):307–318
Gatz SA, Wiesmuller L (2006) p53 in recombination and repair. Cell Death Differ 13(6):1003–1016. doi:10.1038/sj.cdd.4401903
Geisler S, Lonning PE, Aas T et al (2001) Influence of TP53 gene alterations and c-erbB-2 expression on the response to treatment with doxorubicin in locally advanced breast cancer. Cancer Res 61(6):2505–2512
Geisler S, Borresen-Dale AL, Johnsen H et al (2003) TP53 Gene mutations predict the response to neoadjuvant treatment with 5-fluorouracil and mitomycin in locally advanced breast cancer. Clin Cancer Res 9(15):5582–5588
Georges SA, Biery MC, Kim SY et al (2008) Coordinated regulation of cell cycle transcripts by p53-Inducible microRNAs, miR-192 and miR-215. Cancer Res 68(24):10105–10112. doi:10.1158/0008-5472.can-08-1846
Grombacher T, Eichhorn U, Kaina B (1998) p53 is involved in regulation of the DNA repair gene O6-methylguanine-DNA methyltransferase (MGMT) by DNA damaging agents. Oncogene 17(7):845–851. doi:10.1038/sj.onc.1202000
Haldar S, Negrini M, Monne M, Sabbioni S, Croce CM (1994) Down-regulation of bcl-2 by p53 in breast cancer cells. Cancer Res 54(8):2095–2097
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. doi:10.1016/j.cell.2011.02.013
Harper JW, Adami GR, Wei N, Keyomarsi K, Elledge SJ (1993) The p21 Cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 75(4):805–816
Harris LC, Remack JS, Houghton PJ, Brent TP (1996) Wild-type p53 suppresses transcription of the human O6-methylguanine-DNA methyltransferase gene. Cancer Res 56(9):2029–2032
Harris LN, Broadwater G, Lin NU et al (2006) Molecular subtypes of breast cancer in relation to paclitaxel response and outcomes in women with metastatic disease: results from CALGB 9342. Breast Cancer Res BCR 8(6):R66. doi:10.1186/bcr1622
Haupt Y, Maya R, Kazaz A, Oren M (1997) Mdm2 promotes the rapid degradation of p53. Nature 387(6630):296–299. doi:10.1038/387296a0
He L, He X, Lim LP et al (2007a) A microRNA component of the p53 tumour suppressor network. Nature 447(7148):1130–1134. doi:10.1038/nature05939
He L, He X, Lowe SW, Hannon GJ (2007b) microRNAs join the p53 network—another piece in the tumour-suppression puzzle. Nat Rev Cancer 7(11):819–822. doi:10.1038/nrc2232
Heinlein C, Deppert W, Braithwaite AW, Speidel D (2010) A rapid and optimization-free procedure allows the in vivo detection of subtle cell cycle and ploidy alterations in tissues by flow cytometry. Cell Cycle 9(17):3584–3590
Hermeking H, Lengauer C, Polyak K et al (1997) 14-3-3 sigma is a p53-regulated inhibitor of G2/M progression. Mol Cell 1(1):3–11
Hildesheim J, Bulavin DV, Anver MR et al (2002) Gadd45a protects against UV irradiation-induced skin tumors, and promotes apoptosis and stress signaling via MAPK and p53. Cancer Res 62(24):7305–7315
Hinkal G, Parikh N, Donehower LA (2009) Timed somatic deletion of p53 in mice reveals age-associated differences in tumor progression. PLoS ONE 4(8):e6654. doi:10.1371/journal.pone.0006654
Hirose Y, Berger MS, Pieper RO (2001) p53 effects both the duration of G2/M arrest and the fate of temozolomide-treated human glioblastoma cells. Cancer Res 61(5):1957–1963
Hoffman WH, Biade S, Zilfou JT, Chen J, Murphy M (2002) Transcriptional repression of the anti-apoptotic survivin gene by wild type p53. J Biol Chem 277(5):3247–3257. doi:10.1074/jbc.M106643200
Hollander MC, Sheikh MS, Bulavin DV et al (1999) Genomic instability in Gadd45a-deficient mice. Nat Genet 23(2):176–184. doi:10.1038/13802
Hu W, Feng Z, Levine AJ (2012) The regulation of multiple p53 stress responses is mediated through MDM2. Genes Cancer 3(3–4):199–208. doi:10.1177/1947601912454734
Hunten S, Siemens H, Kaller M, Hermeking H (2013) The p53/microRNA network in cancer: experimental and bioinformatics approaches. Adv Exp Med Biol 774:77–101. doi:10.1007/978-94-007-5590-1_5
Hwang BJ, Ford JM, Hanawalt PC, Chu G (1999) Expression of the p48 xeroderma pigmentosum gene is p53-dependent and is involved in global genomic repair. Proc Natl Acad Sci USA 96(2):424–428
Jacks T, Remington L, Williams BO et al (1994) Tumor spectrum analysis in p53-mutant mice. Curr Biol CB 4(1):1–7
Jackson RJ, Adnane J, Coppola D, Cantor A, Sebti SM, Pledger WJ (2002) Loss of the cell cycle inhibitors p21(Cip1) and p27(Kip1) enhances tumorigenesis in knockout mouse models. Oncogene 21(55):8486–8497. doi:10.1038/sj.onc.1205946
Jackson RJ, Engelman RW, Coppola D, Cantor AB, Wharton W, Pledger WJ (2003) p21Cip1 nullizygosity increases tumor metastasis in irradiated mice. Cancer Res 63(12):3021–3025
Janz C, Wiesmuller L (2002) Wild-type p53 inhibits replication-associated homologous recombination. Oncogene 21(38):5929–5933. doi:10.1038/sj.onc.1205757
Jayaraman J, Prives C (1995) Activation of p53 sequence-specific DNA binding by short single strands of DNA requires the p53 C-terminus. Cell 81(7):1021–1029
Jeffers JR, Parganas E, Lee Y et al (2003) Puma is an essential mediator of p53-dependent and -independent apoptotic pathways. Cancer Cell 4(4):321–328
Jiang D, Brady CA, Johnson TM et al (2011) Full p53 transcriptional activation potential is dispensable for tumor suppression in diverse lineages. Proc Natl Acad Sci USA 108(41):17123–17128. doi:10.1073/pnas.1111245108
Kandioler-Eckersberger D, Ludwig C, Rudas M et al (2000) TP53 Mutation and p53 overexpression for prediction of response to neoadjuvant treatment in breast cancer patients. Clin Cancer Res 6(1):50–56
Kandoth C, McLellan MD, Vandin F et al (2013) Mutational landscape and significance across 12 major cancer types. Nature 502(7471):333–339. doi:10.1038/nature12634
Kastan MB, Onyekwere O, Sidransky D, Vogelstein B, Craig RW (1991) Participation of p53 protein in the cellular response to DNA damage. Cancer Res 51(23 Part 1):6304–6311
Kastan MB, Zhan Q, El-Deiry WS et al (1992) A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 71(4):587–597
Kemp CJ, Wheldon T, Balmain A (1994) p53-Deficient mice are extremely susceptible to radiation-induced tumorigenesis. Nat Genet 8(1):66–69. doi:10.1038/ng0994-66
Kern SE, Kinzler KW, Bruskin A et al (1991) Identification of p53 as a sequence-specific DNA-binding protein. Science 252(5013):1708–1711
Khoo KH, Verma CS, Lane DP (2014) Drugging the p53 pathway: understanding the route to clinical efficacy. Nat Rev Drug Discov 13(3):217–236. doi:10.1038/nrd4236
Kracikova M, Akiri G, George A, Sachidanandam R, Aaronson SA (2013) A threshold mechanism mediates p53 cell fate decision between growth arrest and apoptosis. Cell Death Differ 20(4):576–588. doi:10.1038/cdd.2012.155
Kubbutat MH, Jones SN, Vousden KH (1997) Regulation of p53 stability by Mdm2. Nature 387(6630):299–303. doi:10.1038/387299a0
Lane DP (1992) Cancer. p53, guardian of the genome. Nature 358(6381):15–16
Lee S, Elenbaas B, Levine A, Griffith J (1995) p53 and its 14 kDa C-terminal domain recognize primary DNA damage in the form of insertion/deletion mismatches. Cell 81(7):1013–1020
Lee S, Cavallo L, Griffith J (1997) Human p53 binds holliday junctions strongly and facilitates their cleavage. J Biol Chem 272(11):7532–7539
Leu JI, Dumont P, Hafey M, Murphy ME, George DL (2004) Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol 6(5):443–450
Li T, Kon N, Jiang L et al (2012) Tumor suppression in the absence of p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell 149(6):1269–1283. doi:10.1016/j.cell.2012.04.026
Lieber MR, Ma Y, Pannicke U, Schwarz K (2003) Mechanism and regulation of human non-homologous DNA end-joining. Nat Rev Mol Cell Biol 4(9):712–720. doi:10.1038/nrm1202
Liebetrau W, Budde A, Savoia A, Grummt F, Hoehn H (1997) p53 Activates Fanconi anemia group C gene expression. Hum Mol Genet 6(2):277–283
Lin Y, Ma W, Benchimol S (2000) Pidd, a new death-domain-containing protein, is induced by p53 and promotes apoptosis. Nat Genet 26(1):122–127. doi:10.1038/79102
Lin Y, Waldman BC, Waldman AS (2003) Suppression of high-fidelity double-strand break repair in mammalian chromosomes by pifithrin-alpha, a chemical inhibitor of p53. DNA Repair 2(1):1–11
Linke SP, Sengupta S, Khabie N et al (2003) p53 Interacts with hRAD51 and hRAD54, and directly modulates homologous recombination. Cancer Res 63(10):2596–2605
Liu TJ, El-Naggar AK, McDonnell TJ et al (1995) Apoptosis induction mediated by wild-type p53 adenoviral gene transfer in squamous cell carcinoma of the head and neck. Cancer Res 55(14):3117–3122
Liu G, Parant JM, Lang G et al (2004) Chromosome stability, in the absence of apoptosis, is critical for suppression of tumorigenesis in Trp53 mutant mice. Nat Genet 36(1):63–68. doi:10.1038/ng1282
Lloyd DR, Hanawalt PC (2000) p53-dependent global genomic repair of benzo[a]pyrene-7,8-diol-9,10-epoxide adducts in human cells. Cancer Res 60(3):517–521
Lloyd DR, Hanawalt PC (2002) p53 Controls global nucleotide excision repair of low levels of structurally diverse benzo(g)chrysene-DNA adducts in human fibroblasts. Cancer Res 62(18):5288–5294
Lowe SW, Ruley HE, Jacks T, Housman DE (1993a) p53-Dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74(6):957–967
Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T (1993b) p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362(6423):847–849. doi:10.1038/362847a0
Lowe SW, Bodis S, McClatchey A et al (1994) p53 Status and the efficacy of cancer therapy in vivo. Science 266(5186):807–810
Lozano G (2007) The oncogenic roles of p53 mutants in mouse models. Curr Opin Genet Dev 17(1):66–70
Lu X, Bocangel D, Nannenga B, Yamaguchi H, Appella E, Donehower LA (2004) The p53-induced oncogenic phosphatase PPM1D interacts with uracil DNA glycosylase and suppresses base excision repair. Mol Cell 15(4):621–634. doi:10.1016/j.molcel.2004.08.007
Ludwig RL, Bates S, Vousden KH (1996) Differential activation of target cellular promoters by p53 mutants with impaired apoptotic function. Mol Cell Biol 16(9):4952–4960
Malkin D, Li FP, Strong LC et al (1990) Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science 250(4985):1233–1238
Maltzman W, Czyzyk L (1984) UV irradiation stimulates levels of p53 cellular tumor antigen in nontransformed mouse cells. Mol Cell Biol 4(9):1689–1694
Martin-Caballero J, Flores JM, Garcia-Palencia P, Serrano M (2001) Tumor susceptibility of p21(Waf1/Cip1)-deficient mice. Cancer Res 61(16):6234–6238
Massague J (2004) G1 cell-cycle control and cancer. Nature 432(7015):298–306. doi:10.1038/nature03094
Maya R, Balass M, Kim ST et al (2001) ATM-dependent phosphorylation of Mdm2 on serine 395: role in p53 activation by DNA damage. Genes Dev 15(9):1067–1077. doi:10.1101/gad.886901
McDonald ER 3rd, Wu GS, Waldman T, El-Deiry WS (1996) Repair defect in p21WAF1/CIP1-/-human cancer cells. Cancer Res 56(10):2250–2255
Medema RH, Macurek L (2012) Checkpoint control and cancer. Oncogene 31(21):2601–2613. doi:10.1038/onc.2011.451
Meek DW, Anderson CW (2009) Posttranslational modification of p53: cooperative integrators of function. Cold Spring Harb Perspect Biol 1(6):a000950. doi:10.1101/cshperspect.a000950
Michaelis M, Rothweiler F, Barth S et al (2011) Adaptation of cancer cells from different entities to the MDM2 inhibitor nutlin-3 results in the emergence of p53-mutated multi-drug-resistant cancer cells. Cell Death Dis 2:e243
Michaelis M, Rothweiler F, Agha B et al (2012) Human neuroblastoma cells with acquired resistance to the p53 activator RITA retain functional p53 and sensitivity to other p53 activating agents. Cell Death Dis 3:e294. doi:10.1038/cddis.2012.35
Michalak EM, Villunger A, Adams JM, Strasser A (2008) In several cell types tumour suppressor p53 induces apoptosis largely via Puma but Noxa can contribute. Cell Death Differ 15(6):1019–1029. doi:10.1038/cdd.2008.16
Michalak EM, Jansen ES, Happo L et al (2009) Puma and to a lesser extent Noxa are suppressors of Myc-induced lymphomagenesis. Cell Death Differ 16(5):684–696. doi:10.1038/cdd.2008.195
Mihara M, Erster S, Zaika A et al (2003) p53 has a direct apoptogenic role at the mitochondria. Mol Cell 11(3):577–590
Mirza A, McGuirk M, Hockenberry TN et al (2002) Human survivin is negatively regulated by wild-type p53 and participates in p53-dependent apoptotic pathway. Oncogene 21(17):2613–2622. doi:10.1038/sj.onc.1205353
Miyashita T, Reed JC (1995) Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80(2):293–299
Miyashita T, Harigai M, Hanada M, Reed JC (1994) Identification of a p53-dependent negative response element in the bcl-2 gene. Cancer Res 54(12):3131–3135
Momand J, Zambetti GP, Olson DC, George D, Levine AJ (1992) The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53-mediated transactivation. Cell 69(7):1237–1245
Moroni MC, Hickman ES, Lazzerini Denchi E et al (2001) Apaf-1 is a transcriptional target for E2F and p53. Nat Cell Biol 3(6):552–558. doi:10.1038/35078527
Muller M, Wilder S, Bannasch D et al (1998) p53 Activates the CD95 (APO-1/Fas) gene in response to DNA damage by anticancer drugs. J Exp Med 188(11):2033–2045
Myung K, Braastad C, He DM, Hendrickson EA (1998) KARP-1 is induced by DNA damage in a p53- and ataxia telangiectasia mutated-dependent fashion. Proc Natl Acad Sci USA 95(13):7664–7669
Nakano K, Vousden KH (2001) PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell 7(3):683–694
Nakano K, Balint E, Ashcroft M, Vousden KH (2000) A ribonucleotide reductase gene is a transcriptional target of p53 and p73. Oncogene 19(37):4283–4289
Niculescu AB 3rd, Chen X, Smeets M, Hengst L, Prives C, Reed SI (1998) Effects of p21(Cip1/Waf1) at both the G1/S and the G2/M cell cycle transitions: pRb is a critical determinant in blocking DNA replication and in preventing endoreduplication. Mol Cell Biol 18(1):629–643
Oda E, Ohki R, Murasawa H et al (2000a) Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 288(5468):1053–1058
Oda K, Arakawa H, Tanaka T et al (2000b) p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102(6):849–862
Offer H, Wolkowicz R, Matas D, Blumenstein S, Livneh Z, Rotter V (1999) Direct involvement of p53 in the base excision repair pathway of the DNA repair machinery. FEBS Lett 450(3):197–204
Offer H, Milyavsky M, Erez N et al (2001a) Structural and functional involvement of p53 in BER in vitro and in vivo. Oncogene 20(5):581–589. doi:10.1038/sj.onc.1204120
Offer H, Zurer I, Banfalvi G et al (2001b) p53 Modulates base excision repair activity in a cell cycle-specific manner after genotoxic stress. Cancer Res 61(1):88–96
Ogryzko VV, Wong P, Howard BH (1997) WAF1 retards S-phase progression primarily by inhibition of cyclin-dependent kinases. Mol Cell Biol 17(8):4877–4882
Ohki R, Nemoto J, Murasawa H et al (2000) Reprimo, a new candidate mediator of the p53-mediated cell cycle arrest at the G2 phase. J Biol Chem 275(30):22627–22630. doi:10.1074/jbc.C000235200
Okorokov AL, Warnock L, Milner J (2002) Effect of wild-type, S15D and R175H p53 proteins on DNA end joining in vitro: potential mechanism of DNA double-strand break repair modulation. Carcinogenesis 23(4):549–557
Oliner JD, Pietenpol JA, Thiagalingam S, Gyuris J, Kinzler KW, Vogelstein B (1993) Oncoprotein MDM2 conceals the activation domain of tumour suppressor p53. Nature 362(6423):857–860. doi:10.1038/362857a0
Olivier M, Hollstein M, Hainaut P (2010) TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb Perspect Biol 2(1):a001008. doi:10.1101/cshperspect.a001008
Otsuka K, Ochiya T (2014) Genetic networks lead and follow tumor development: microRNA regulation of cell cycle and apoptosis in the p53 pathways. BioMed Res Int 2014:749724. doi:10.1155/2014/749724
Owen-Schaub LB, Zhang W, Cusack JC et al (1995) Wild-type human p53 and a temperature-sensitive mutant induce Fas/APO-1 expression. Mol Cell Biol 15(6):3032–3040
Passalaris TM, Benanti JA, Gewin L, Kiyono T, Galloway DA (1999) The G(2) checkpoint is maintained by redundant pathways. Mol Cell Biol 19(9):5872–5881
Petitjean A, Achatz MI, Borresen-Dale AL, Hainaut P, Olivier M (2007) TP53 Mutations in human cancers: functional selection and impact on cancer prognosis and outcomes. Oncogene 26(15):2157–2165
Polyak K, Waldman T, He TC, Kinzler KW, Vogelstein B (1996) Genetic determinants of p53-induced apoptosis and growth arrest. Genes Dev 10(15):1945–1952
Purdie CA, Harrison DJ, Peter A et al (1994) Tumour incidence, spectrum and ploidy in mice with a large deletion in the p53 gene. Oncogene 9(2):603–609
Rafferty JA, Clarke AR, Sellappan D, Koref MS, Frayling IM, Margison GP (1996) Induction of murine O6-alkylguanine-DNA-alkyltransferase in response to ionising radiation is p53 gene dose dependent. Oncogene 12(3):693–697
Raver-Shapira N, Marciano E, Meiri E et al (2007) Transcriptional activation of miR-34a contributes to p53-mediated apoptosis. Mol Cell 26(5):731–743. doi:10.1016/j.molcel.2007.05.017
Reed M, Woelker B, Wang P, Wang Y, Anderson ME, Tegtmeyer P (1995) The C-terminal domain of p53 recognizes DNA damaged by ionizing radiation. Proc Natl Acad Sci USA 92(21):9455–9459
Roberts SA, Gordenin DA (2014) Hypermutation in human cancer genomes: footprints and mechanisms. Nat Rev Cancer 14(12):786–800. doi:10.1038/nrc3816
Robles AI, Bemmels NA, Foraker AB, Harris CC (2001) APAF-1 is a transcriptional target of p53 in DNA damage-induced apoptosis. Cancer Res 61(18):6660–6664
Rouault JP, Falette N, Guehenneux F et al (1996) Identification of BTG2, an antiproliferative p53-dependent component of the DNA damage cellular response pathway. Nat Genet 14(4):482–486. doi:10.1038/ng1296-482
Rowan S, Ludwig RL, Haupt Y et al (1996) Specific loss of apoptotic but not cell-cycle arrest function in a human tumor derived p53 mutant. EMBO J 15(4):827–838
Ryu MS, Lee MS, Hong JW, Hahn TR, Moon E, Lim IK (2004) TIS21/BTG2/PC3 is expressed through PKC-delta pathway and inhibits binding of cyclin B1-Cdc2 and its activity, independent of p53 expression. Exp Cell Res 299(1):159–170. doi:10.1016/j.yexcr.2004.05.014
Scherer SJ, Welter C, Zang KD, Dooley S (1996) Specific in vitro binding of p53 to the promoter region of the human mismatch repair gene hMSH2. Biochem Biophys Res Commun 221(3):722–728. doi:10.1006/bbrc.1996.0663
Scherer SJ, Maier SM, Seifert M et al (2000) p53 and c-Jun functionally synergize in the regulation of the DNA repair gene hMSH2 in response to UV. J Biol Chem 275(48):37469–37473. doi:10.1074/jbc.M006990200
Schlereth K, Beinoraviciute-Kellner R, Zeitlinger MK et al (2010) DNA binding cooperativity of p53 modulates the decision between cell-cycle arrest and apoptosis. Mol Cell 38(3):356–368. doi:10.1016/j.molcel.2010.02.037
Schmitt CA, Fridman JS, Yang M et al (2002) A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell 109(3):335–346
Scoumanne A, Cho SJ, Zhang J, Chen X (2011) The cyclin-dependent kinase inhibitor p21 is regulated by RNA-binding protein PCBP4 via mRNA stability. Nucleic Acids Res 39(1):213–224. doi:10.1093/nar/gkq778
Sengupta S, Harris CC (2005) p53: traffic cop at the crossroads of DNA repair and recombination. Nat Rev Mol Cell Biol 6(1):44–55. doi:10.1038/nrm1546
Seo YR, Fishel ML, Amundson S, Kelley MR, Smith ML (2002) Implication of p53 in base excision DNA repair: in vivo evidence. Oncogene 21(5):731–737. doi:10.1038/sj.onc.1205129
Shao L, Sun Y, Zhang Z et al (2010) Deletion of proapoptotic Puma selectively protects hematopoietic stem and progenitor cells against high-dose radiation. Blood 115(23):4707–4714. doi:10.1182/blood-2009-10-248872
Shaw P, Bovey R, Tardy S, Sahli R, Sordat B, Costa J (1992) Induction of apoptosis by wild-type p53 in a human colon tumor-derived cell line. Proc Natl Acad Sci USA 89(10):4495–4499
Shieh SY, Ikeda M, Taya Y, Prives C (1997) DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91(3):325–334
Slatter TL, Ganesan P, Holzhauer C et al (2010) p53-Mediated apoptosis prevents the accumulation of progenitor B cells and B-cell tumors. Cell Death Differ 17(3):540–550
Smith ML, Chen IT, Zhan Q et al (1994) Interaction of the p53-regulated protein Gadd45 with proliferating cell nuclear antigen. Science 266(5189):1376–1380
Smith ML, Chen IT, Zhan Q, O’Connor PM, Fornace AJ Jr (1995) Involvement of the p53 tumor suppressor in repair of u.v.-type DNA damage. Oncogene 10(6):1053–1059
Smith ML, Ford JM, Hollander MC et al (2000) p53-Mediated DNA repair responses to UV radiation: studies of mouse cells lacking p53, p21, and/or gadd45 genes. Mol Cell Biol 20(10):3705–3714
Speidel D (2010) Transcription-independent p53 apoptosis: an alternative route to death. Trends Cell Biol 20(1):14–24
Speidel D, Helmbold H, Deppert W (2006) Dissection of transcriptional and non-transcriptional p53 activities in the response to genotoxic stress. Oncogene 25(6):940–953
Spurgers KB, Gold DL, Coombes KR et al (2006) Identification of cell cycle regulatory genes as principal targets of p53-mediated transcriptional repression. J Biol Chem 281(35):25134–25142. doi:10.1074/jbc.M513901200
St Clair S, Giono L, Varmeh-Ziaie S et al (2004) DNA damage-induced downregulation of Cdc25C is mediated by p53 via two independent mechanisms: one involves direct binding to the cdc25C promoter. Mol Cell 16(5):725–736. doi:10.1016/j.molcel.2004.11.002
Stewart N, Hicks GG, Paraskevas F, Mowat M (1995) Evidence for a second cell cycle block at G2/M by p53. Oncogene 10(1):109–115
Stivala LA, Riva F, Cazzalini O, Savio M, Prosperi E (2001) p21(waf1/cip1)-null human fibroblasts are deficient in nucleotide excision repair downstream the recruitment of PCNA to DNA repair sites. Oncogene 20(5):563–570. doi:10.1038/sj.onc.1204132
Sturzbecher HW, Donzelmann B, Henning W, Knippschild U, Buchhop S (1996) p53 is linked directly to homologous recombination processes via RAD51/RecA protein interaction. EMBO J 15(8):1992–2002
Sugrue MM, Shin DY, Lee SW, Aaronson SA (1997) Wild-type p53 triggers a rapid senescence program in human tumor cells lacking functional p53. Proc Natl Acad Sci USA 94(18):9648–9653
Suzuki K, Mori I, Nakayama Y, Miyakoda M, Kodama S, Watanabe M (2001) Radiation-induced senescence-like growth arrest requires TP53 function but not telomere shortening. Radiat Res 155(1 Pt 2):248–253
Tanaka H, Arakawa H, Yamaguchi T et al (2000) A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature 404(6773):42–49. doi:10.1038/35003506
Tang HY, Zhao K, Pizzolato JF, Fonarev M, Langer JC, Manfredi JJ (1998) Constitutive expression of the cyclin-dependent kinase inhibitor p21 is transcriptionally regulated by the tumor suppressor protein p53. J Biol Chem 273(44):29156–29163
Tang W, Willers H, Powell SN (1999) p53 Directly enhances rejoining of DNA double-strand breaks with cohesive ends in gamma-irradiated mouse fibroblasts. Cancer Res 59(11):2562–2565
Tarasov V, Jung P, Verdoodt B et al (2007) Differential regulation of microRNAs by p53 revealed by massively parallel sequencing: miR-34a is a p53 target that induces apoptosis and G1-arrest. Cell Cycle 6(13):1586–1593
Taylor WR, DePrimo SE, Agarwal A et al (1999) Mechanisms of G2 arrest in response to overexpression of p53. Mol Biol Cell 10(11):3607–3622
Tchelebi L, Ashamalla H, Graves PR (2014) Mutant p53 and the response to chemotherapy and radiation. Sub-cell Biochem 85:133–159. doi:10.1007/978-94-017-9211-0_8
Therrien JP, Drouin R, Baril C, Drobetsky EA (1999) Human cells compromised for p53 function exhibit defective global and transcription-coupled nucleotide excision repair, whereas cells compromised for pRb function are defective only in global repair. Proc Natl Acad Sci USA 96(26):15038–15043
Timofeev O, Schlereth K, Wanzel M et al (2013) p53 DNA binding cooperativity is essential for apoptosis and tumor suppression in vivo. Cell Rep 3(5):1512–1525. doi:10.1016/j.celrep.2013.04.008
Tront JS, Hoffman B, Liebermann DA (2006) Gadd45a suppresses Ras-driven mammary tumorigenesis by activation of c-Jun NH2-terminal kinase and p38 stress signaling resulting in apoptosis and senescence. Cancer Res 66(17):8448–8454. doi:10.1158/0008-5472.can-06-2013
Tront JS, Huang Y, Fornace AJ Jr, Hoffman B, Liebermann DA (2010) Gadd45a functions as a promoter or suppressor of breast cancer dependent on the oncogenic stress. Cancer Res 70(23):9671–9681. doi:10.1158/0008-5472.can-10-2177
Utrera R, Collavin L, Lazarevic D, Delia D, Schneider C (1998) A novel p53-inducible gene coding for a microtubule-localized protein with G2-phase-specific expression. EMBO J 17(17):5015–5025. doi:10.1093/emboj/17.17.5015
Valente LJ, Gray DH, Michalak EM et al (2013) p53 Efficiently suppresses tumor development in the complete absence of its cell-cycle inhibitory and proapoptotic effectors p21, Puma, and Noxa. Cell Rep 3(5):1339–1345. doi:10.1016/j.celrep.2013.04.012
Villunger A, Michalak EM, Coultas L et al (2003) p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science 302(5647):1036–1038
Vogelstein B, Lane D, Levine AJ (2000) Surfing the p53 network. Nature 408(6810):307–310
Vousden KH (2006) Outcomes of p53 activation—spoilt for choice. J Cell Sci 119(Pt 24):5015–5020
Vousden KH, Lane DP (2007) p53 in health and disease. Nat Rev Mol Cell Biol 8(4):275–283
Vousden KH, Prives C (2009) Blinded by the light: the Growing Complexity of p53. Cell 137(3):413–431
Waga S, Hannon GJ, Beach D, Stillman B (1994) The p21 inhibitor of cyclin-dependent kinases controls DNA replication by interaction with PCNA. Nature 369(6481):574–578. doi:10.1038/369574a0
Waldman T, Kinzler KW, Vogelstein B (1995) p21 is necessary for the p53-mediated G1 arrest in human cancer cells. Cancer Res 55(22):5187–5190
Wang XW, Yeh H, Schaeffer L et al (1995) p53 Modulation of TFIIH-associated nucleotide excision repair activity. Nat Genet 10(2):188–195. doi:10.1038/ng0695-188
Wang XW, Zhan Q, Coursen JD et al (1999) GADD45 induction of a G2/M cell cycle checkpoint. Proc Natl Acad Sci USA 96(7):3706–3711
Wani MA, Zhu Q, El-Mahdy M, Venkatachalam S, Wani AA (2000) Enhanced sensitivity to anti-benzo(a)pyrene-diol-epoxide DNA damage correlates with decreased global genomic repair attributable to abrogated p53 function in human cells. Cancer Res 60(8):2273–2280
Weinert TA, Hartwell LH (1988) The RAD9 gene controls the cell cycle response to DNA damage in Saccharomyces cerevisiae. Science 241(4863):317–322
Wu GS, Burns TF, McDonald ER 3rd et al (1997) KILLER/DR5 is a DNA damage-inducible p53-regulated death receptor gene. Nat Genet 17(2):141–143. doi:10.1038/ng1097-141
Xiong Y, Hannon GJ, Zhang H, Casso D, Kobayashi R, Beach D (1993) p21 is a universal inhibitor of cyclin kinases. Nature 366(6456):701–704. doi:10.1038/366701a0
Xue L, Zhou B, Liu X, Qiu W, Jin Z, Yen Y (2003) Wild-type p53 regulates human ribonucleotide reductase by protein—protein interaction with p53R2 as well as hRRM2 subunits. Cancer Res 63(5):980–986
Yang T, Namba H, Hara T et al (1997) p53 Induced by ionizing radiation mediates DNA end-jointing activity, but not apoptosis of thyroid cells. Oncogene 14(13):1511–1519. doi:10.1038/sj.onc.1200979
Yonish-Rouach E, Resnitzky D, Lotem J, Sachs L, Kimchi A, Oren M (1991) Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited by interleukin-6. Nature 352(6333):345–347. doi:10.1038/352345a0
Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B (2001) PUMA induces the rapid apoptosis of colorectal cancer cells. Mol Cell 7(3):673–682
Yu J, Wang Z, Kinzler KW, Vogelstein B, Zhang L (2003) PUMA mediates the apoptotic response to p53 in colorectal cancer cells. Proc Natl Acad Sci USA 100(4):1931–1936. doi:10.1073/pnas.2627984100
Yu H, Shen H, Yuan Y et al (2010) Deletion of Puma protects hematopoietic stem cells and confers long-term survival in response to high-dose gamma-irradiation. Blood 115(17):3472–3480. doi:10.1182/blood-2009-10-248278
Zhan Q, Antinore MJ, Wang XW et al (1999) Association with Cdc2 and inhibition of Cdc2/Cyclin B1 kinase activity by the p53-regulated protein Gadd45. Oncogene 18(18):2892–2900. doi:10.1038/sj.onc.1202667
Zhou J, Prives C (2003) Replication of damaged DNA in vitro is blocked by p53. Nucleic Acids Res 31(14):3881–3892
Zhou J, Ahn J, Wilson SH, Prives C (2001) A role for p53 in base excision repair. EMBO J 20(4):914–923. doi:10.1093/emboj/20.4.914
Zhou G, Wang J, Zhao M et al (2014) Gain-of-function mutant p53 promotes cell growth and cancer cell metabolism via inhibition of AMPK activation. Mol Cell 54(6):960–974. doi:10.1016/j.molcel.2014.04.024
Zhu J, Chen X (2000) MCG10, a novel p53 target gene that encodes a KH domain RNA-binding protein, is capable of inducing apoptosis and cell cycle arrest in G(2)-M. Mol Cell Biol 20(15):5602–5618
Zurer I, Hofseth LJ, Cohen Y et al (2004) The role of p53 in base excision repair following genotoxic stress. Carcinogenesis 25(1):11–19. doi:10.1093/carcin/bgg186
Acknowledgments
Work on DNA damage responses in my laboratory has been supported by Grants from Cure Cancer Australia Foundation, Cancer Institute New South Wales, Anthony Rothe Memorial Trust and Tour de Cure.
Conflict of interest
The author declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Speidel, D. The role of DNA damage responses in p53 biology. Arch Toxicol 89, 501–517 (2015). https://doi.org/10.1007/s00204-015-1459-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1459-z