Abstract
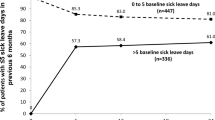
The aim of this noninterventional study (NIS) was to analyze the changes in sickness absence, disease activity, and functional capacity in employed rheumatoid arthritis (RA) patients during adalimumab treatment. RA patients receiving adalimumab according to label instructions (40 mg every other week) were evaluated at regular intervals in a multicenter prospective NIS. Patients provided information on sickness absence in the 12 months preceding treatment initiation (baseline) and at months 6 and 12. Disease activity was assessed by the Disease Activity Score using 28 joints, and physical function was assessed via the Hannover Functional Ability Questionnaire, a patient self-questionnaire comparable with the Health Assessment Questionnaire-Disability Index. We present data on 1,157 patients who were employed (part time or full time) at baseline. Patients were categorized by the length of sickness absence at baseline. At baseline, patients with absences of 6 weeks or more in the previous year (n = 226 [19.5%]) accounted for 77% of the documented weeks of sickness absence, and patients with absences of more than 12 weeks (n = 98 [8.5%]) accounted for 54% of sickness absence weeks. During 12 months of adalimumab treatment, disease activity decreased, functional capacity improved, and sickness absence was reduced. The greatest decrease in sickness absence was observed in patients with more than 12 weeks of sick leave in the year prior to adalimumab therapy. These patients also showed gains in function comparable with those observed in other employed patients. We conclude that sustaining and improving functional capacity represent the key to preservation of work capability.



Similar content being viewed by others
References
Smolen JS, Aletaha D (2009) Developments in the clinical understanding of rheumatoid arthritis. Arthr Res Ther 11:204. doi:10.1186/ar2535
Hulseman JL, Ruof J, Zeidler H, Mittendor T (2006) Costs in rheumatology: results and lessons learned from the ‘Hannover Costing Study’. Rheumatol Int 26:704–711. doi:10.1007/s00296-005-0070-7
Huscher D, Merkesdal S, Thiele K, Zeidler H, Schneider M, Zink A (2006) Cost of illness in rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and systemic lupus erythematosus in Germany. Ann Rheum Dis 65:1175–1183. doi:10.1136/ard.2005.046367
Mau W, Beyer W, Ehlebrach-König I, Engel M, Genth E, Greitemann B, Jäckel WH, Zink A (2008) Burden of illness: first routine report on socio medical consequences of inflammatory rheumatic disease in Germany. Z Rheumatol 67:157–164. doi:10.1007/s00393-007-0250-9 (German)
Zink A, Mau W, Schneider M (2001) Epidemiological and socio medical aspects of inflammatory rheumatic systemic diseases. Internist 42:211–222 (German)
Burmester GR, Mariette X, Montecucco C, Monteagudo-Sáez I, Malaise M, Tzioufas AG, Bijlsma JWJ, Unnebrink K, Kary S, Kupper H (2007) Adalimumab alone and in combination with disease-modifying antirheumatic drugs for the treatment of rheumatoid arthritis in clinical practice: the Research in Active Rheumatoid Arthritis (ReAct) trial. Ann Rheum Dis 66:732–739. doi:10.1136/ard.2006.066761
Kirchoff T, Ruof J, Mittendorf T, Rihl M, Bertnateck M, Mau W, Zeidler H, Schmidt RE, Merkesdal S (2011) Cost of illness in rheumatoid arthritis in Germany in 1997–1998 and 2002: cost drivers and cost savings. Rheumatology 20:756–761. doi:10.1093/rheumatology/keq398
Silverman SL (2009) From randomized controlled trials to observational studies. Am J Med 122:120–144. doi:10.1016/j.amjmed.2008.09.030
Kleinert S, Tony H-P, Krause A et al (2011) Impact of patient and disease characteristics on therapeutic success during adalimumab treatment of patients with rheumatoid arthritis: data from a German non-interventional observational study. Rheumatol Int. doi:10.1007/s00296-011-2033-5 (Epub ahead of print)
Vander Cruyssen B, Van Looy S, Wyns B, Westhovens R, Durez P, Van den Bosch F, Veys EM, Mielants H, De Clerck L, Peretz A, Malaise M, Verbruggen L, Vastesaeger N, Geldhof A, Boullart L, De Keyser F (2005) DAS28 best reflects the physician’s clinical judgment of response to infliximab therapy in rheumatoid patients: validation of the DAS28 score in patients under infliximab treatment. Arthr Res Ther 7:R1063–R1071. doi:10.1186/ar1787
Lautenschlaeger J, Mau W, Kohlmann T, Raspe HH, Struve F, Bruckle W, Zeidler H (1997) Comparative evaluation of a German version of the Health Assessment Questionnaire (HAQ) and the Hannover Functional Status Questionnaire (HFSQ). Z Rheumatol 56:144–151 (German)
Raspe HH, Hagedorn U, Kohlmann T, Mattussek S (1990) The Hannover Functional Status Questionnaire (HFSQ): an instrument for functional diagnostic in polyarticular joint disease. In: Siegrist J (ed) Wohnortnahe Betreuung Rheumakranker. Ergebnisse sozialwissenschaftlicher Evaluation eines Modellversuchs. Schattauer, Stuttgart, pp 164–182 (German)
Greenhouse SW, Geisser S (1959) On methods in the analysis of profile data. Psychometrika 24:95–112
Bejarano V, Quinn M, Conaghan PG, Reece R, Keenan AM, Walker D, Gough A, Green M, McGonagle D, Adebajo A, Jarrett S, Doherty S, Hordon L, Melsom R, Unnebrink K, Kupper H, Emery P (2008) Effect of the early use of the anti-tumor necrosis factor adalimumab on the prevention of job loss in patients with early rheumatoid arthritis. Arthr Rheum 59:1467–1474. doi:10.1002/art.24106
Augustsson J, Neovius M, Cullinane-Carli C, Eksborg S, van Vollenhofen RF (2010) Rheumatoid arthritis patients treated with TNF antagonists increase their participation in the work-force–potential for significant long-term indirect cost gains. Data from a population-based registry. Ann Rheum Dis 69:126–131. doi:10.1136/ard.2009.108035
Merkesdal S, Ruof J, Huelsemann JL, Mittendorf T, Handelmann S, Mau W, Zeidler H (2005) Indirect cost assessment in patients with rheumatoid arthritis (RA): comparison of data from the health-economic patient questionnaire HEQ-RA and insurance claims data. Arthr Rheum 53:234–240. doi:10.1002/art.21080
Severens JL, Mulder J, Laheij RJF, Verbeek ALM (2000) Precision and accuracy in measuring absence from work as a basis for calculating productivity costs in the Netherlands. Soc Sci Med 51:243–249
Schneider J, Beblo M (2010) Health at work—indicators and determinants. A revised literature and data review for Germany. http://doku.iab.de/discussionpapers/2010/dp1710.pdf. Accessed 01 Mar 2011
Aletaha D, Ward MM (2006) Duration of rheumatoid arthritis influences the degree of functional improvement in clinical trials. Ann Rheum Dis 65:227–233. doi:10.1136/ard.2005.038513
Acknowledgments
This study and the writing of this paper were funded by Abbott GmbH & Co. KG. We thank Britta Eichhorn, MA, and Sharon L. Cross, PhD, for providing medical writing services on behalf of Abbott GmbH & Co. KG.
Conflict of Interest
K. Krüger, J. Wollenhaupt, H.M. Lorenz, and E. Röther received honoraria, speaker’s fees, or compensation for consultation from Abbott GmbH & Co. KG. B. M. Wittig is an employee of Abbott.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krüger, K., Wollenhaupt, J., Lorenz, HM. et al. Reduction in sickness absence in patients with rheumatoid arthritis receiving adalimumab: data from a German noninterventional study. Rheumatol Int 32, 3977–3983 (2012). https://doi.org/10.1007/s00296-011-2317-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-011-2317-9