Abstract
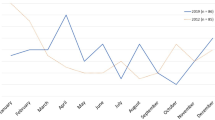
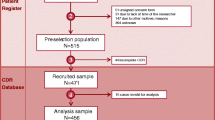
Acute gastroenteritis (AGE) caused by rotavirus (RV) is a common disease among infants and toddlers, often leading to hospitalization and, in resource-poor countries, to death. However, little is known on specific complications of severe RV-positive (RV+) AGE and on the clinical course in chronically ill children. This was a retrospective analysis of data for children <5 years of age hospitalized due to AGE during six rotavirus seasons in three large German pediatric hospitals. The primary study end point was the incidence and type of complications in RV+ versus RV-negative (RV−) cases. A total of 6,884 episodes of AGE in hospitalized children aged <5 years were included in this analysis. Of the 4,880 stools tested for RV, 2,118 (43.4%) were RV+. Hypernatremia was significantly more common in RV+ AGE (P < 0.001) and was associated with severe disease, need for intensive care treatment, and longer duration of hospitalization. Metabolic disorders, particularly hypoglycemia, were more common in RV+ AGE. In contrast, symptoms such as respiratory infections, neurological, and abdominal symptoms were more common in children with RV− AGE. Conclusions: hypernatremia is a specific complication of RV+ AGE. RV would therefore appear to be the cause of infant toxicosis, the traditional descriptive term for severe dehydration and clinical deterioration following AGE.


Similar content being viewed by others
References
Adler A, Wollach B, Kilman A, Gottesman G (2005) Enteric gram-negative sepsis complicating rotavirus gastroenteritis in previously healthy infants. Clin Pediatr (Phila) 44:351–354
Anonymous (1992) Recommendations for composition of oral rehydration solutions for the children of Europe. Report of an ESPGAN Working Group. J Pediatr Gastroenterol Nutr 14(1):113–115
Caraballo RH, Gañez L, Santos Cde L et al (2009) Benign infantile seizures with mild gastroenteritis: study of 22 patients. Seizure 18(10):686–689
Cicchetti R, Iacobini M, Midulla F, Papoff P, Mancuso M, Moretti C (2006) Pantoea agglomerans sepsis after rotavirus gastroenteritis. Pediatr Infect Dis J 25:280–281
Ciftçi E, Tapisiz A, Ozdemir H et al (2009) Bacteraemia and candidaemia: a considerable and underestimated complication of severe rotavirus gastroenteritis. Scand J Infect Dis 41(11–12):857–861
de Villiers FP, Steele AD, Driessen M (2003) Central nervous system involvement in neonatal rotavirus infection. Ann Trop Paediatr 23(4):309–312
Dickey M, Jamison L, Michaud L et al (2009) Rotavirus meningoencephalitis in a previously healthy child and a review of the literature. Pediatr Infect Dis J 28(4):318–321
Festini F, Cocchi P, Mambretti D et al (2010) Nosocomial rotavirus gastroenteritis in pediatric patients: a multi-center prospective cohort study. BMC Infect Dis 10:235
Fitts SW, Green M, Reyes J et al (1995) Clinical features of nosocomial rotavirus infection in pediatric liver transplant recipients. Clin Transplant 9(3 Pt 1):201–204
Forster J, Guarino A, Parez N et al (2009) Hospital-based surveillance to estimate the burden of rotavirus gastroenteritis among European children younger than 5 years of age. Pediatrics 123:e393–e400
Giaquinto C, Van Damme P, Huet F et al (2007) Clinical consequences of rotavirus acute gastroenteritis in Europe, 2004–2005: the REVEAL study. J Infect Dis 195(Suppl 1):S26–S35
Gimenez-Sanchez F, Delgado-Rubio A, Martinon-Torres F et al (2010) Multicenter prospective study analysing the role of rotavirus on acute gastroenteritis in Spain. Acta Paediatr 99(5):738–742
Grimwood K, Lambert SB (2009) Rotavirus vaccines: opportunities and challenges. Hum Vaccin 5:57–69
Hung JJ, Wen HY, Yen MH et al (2003) Rotavirus gastroenteritis associated with afebrile convulsion in children: clinical analysis of 40 cases. Chang Gung Med J 26:654–659
Huppertz HI, Salman N, Giaquinto C (2008) Risk factors for severe rotavirus gastroenteritis. Pediatr Infect Dis J 27:S11–S19
Iturriza-Gómara M, Auchterlonie IA, Zaw W et al (2002) Rotavirus gastroenteritis and central nervous system (CNS) infection: characterization of the VP7 and VP4 genes of rotavirus strains isolated from paired fecal and cerebrospinal fluid samples from a child with CNS disease. J Clin Microbiol 40(12):4797–4799
Iyadurai S, Troester M, Harmala J, Bodensteiner J (2007) Benign afebrile seizures in acute gastroenteritis: is rotavirus the culprit? J Child Neurol 22:887–890
Jacobson J, Bohn D (1993) Severe hypernatremic dehydration and hyperkalemia in an infant with gastroenteritis secondary to rotavirus. Ann Emerg Med 22(10):1630–1632
Johansen K, Hedlund KO, Zweygberg-Wirgart B, Bennet R (2008) Complications attributable to rotavirus-induced diarrhoea in a Swedish paediatric population: report from an 11-year surveillance. Scand J Inf Dis 40(11–12):958–964
Karstens C, Baumgarte S, Friedrich AW et al (2009) Incidence and risk factors for community-acquired acute gastroenteritis in north-west Germany in 2004. Eur J Clin Microbiol Infect Dis 28:935–943
Kashiwagi Y, Sato S, Nakamura M et al (2007) Klebsiella oxytoca septicemia complicating rotavirus-associated acute diarrhea. Pediatr Infect Dis J 26:191–192
Keller KM, Schmidt H, Wirth S, Queisser-Luft A, Schumacher R (1991) Differences in the clinical and radiologic patterns of rotavirus and non-rotavirus necrotizing enterocolitis. Pediatr Infect Dis J 10:734–738
Linhares AC, Velázquez FR, Pérez-Schael I et al (2008) Efficacy and safety of an oral live attenuated human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in Latin American infants: randomised, double-blind controlled study. Lancet 371:1181–1189
Liu B, Fujita Y, Arakawa C et al (2009) Detection of rotavirus RNA and antigens in serum and cerebrospinal fluid samples from diarrheic children with seizures. Jpn J Infect Dis 62(4):279–283
Longmore DK, Batch JA, McMahon SK, Conwell LS (2010) Klebsiella pneumoniae bacteraemia complicating rotavirus gastroenteritis in two infants with glucocorticoid deficiency. J Pediatr Endocrinol Metab 23(3):293–295
Lowenthal A, Livni G, Amir J, Samra Z, Ashkenazi S (2006) Secondary bacteremia after rotavirus gastroenteritis in infancy. Pediatrics 117:224–226
Makino M, Tanabe Y, Shinozaki K, Matsuno S, Furuya T (1996) Haemorrhagic shock and encephalopathy associated with rotavirus infection. Acta Paediatr 85:632–634
Martin A, Batty A, Roberts JA, Standaert B (2009) Cost-effectiveness of infant vaccination with RIX4414 (Rotarix) in the UK. Vaccine 27(33):4520–4528
Mel E, Nahum E, Lowenthal A, Ashkenazi S (2006) Extended spectrum beta-lactamase-positive Escherichia coli bacteremia complicating rotavirus gastroenteritis. Pediatr Infect Dis J 25:962
Narchi H (2004) Benign afebrile cluster convulsions with gastroenteritis: an observational study. BMC Pediatr 4:2
Parashar UD, Hummelman EG, Bresee JS, Miller MA, Glass RI (2003) Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis 9:565–572
Pediatric ROTavirus European CommitTee (PROTECT) (2006) The paediatric burden of rotavirus disease in Europe. Epidemiol Infect 134:908–916
Phua KB, Lim FS, Lau YL et al (2009) Safety and efficacy of human rotavirus vaccine during the first 2 years of life in Asian infants: randomised, double-blind, controlled study. Vaccine 27:5936–5941
Price TG, Kallenborn JC (2000) Infant hypernatremia: a case report. J Emerg Med 19(2):153–157
Ramig RF (2007) Systemic rotavirus infection. Expert Rev Anti Infect Ther 5:591–612
Rayani A, Bode U, Habas E et al (2007) Rotavirus infections in paediatric oncology patients: a matched-pairs analysis. Scand J Gastroenterol 42(1):81–87
Riedel F, Kroener T, Stein K, Nuesslein TG, Rieger CH (1996) Rotavirus infection and bradycardia–apnoea-episodes in the neonate. Eur J Pediatr 155:36–40
Ruíz-Palacios GM, Pérez-Schael I, Velázquez FR et al (2006) Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med 354:11–22
Schulte S (1985) Lehrbuch der Kinderheilkunde. Fischer Verlag, Stuttgart, p 133
Schumacher RF, Forster J (1999) The CNS symptoms of rotavirus infections under the age of two. Klin Padiatr 211(2):61–64
Sharma R, Garrison RD, Tepas JJ 3rd et al (2004) Rotavirus-associated necrotizing enterocolitis: an insight into a potentially preventable disease? J Pediatr Surg 39:453–457
Standaert B, Parez N, Tehard B, Colin X, Detournay B (2008) Cost-effectiveness analysis of vaccination against rotavirus with RIX4414 in France. Appl Health Econ Health Policy 6(4):199–216
Stelzmueller I, Wiesmayr S, Eller M et al (2007) Enterocolitis due to simultaneous infection with rotavirus and Clostridium difficile in adult and pediatric solid organ transplantation. J Gastrointest Surg 11:911–917
Stelzmueller I, Wiesmayr S, Swenson BR et al (2007) Rotavirus enteritis in solid organ transplant recipients: an underestimated problem? Transpl Infect Dis 9:281–285
Vesikari T, Itzler R, Karvonen A et al (2009) RotaTeq®, a pentavalent rotavirus vaccine: efficacy and safety among infants in Europe. Vaccine 28:345–351
Vesikari T, Karvonen A, Prymula R et al (2007) Efficacy of human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in European infants: randomised, double-blind controlled study. Lancet 370:1757–1763
Vesikari T, Matson DO, Dennehy P et al (2006) Safety and efficacy of a pentavalent human–bovine (WC3) reassortant rotavirus vaccine. N Engl J Med 354:23–33
Wiese-Posselt M, Matysiak-Klose D, Gilsdorf A et al (2007) Rotaviren in Deutschland. Monatsschr Kinderheilkd 155:167–175
World Health Organization (2007) International statistical classification of diseases and related health problems, 10th revision. http://apps.who.int/classifications/apps/icd/icd10online/. Accessed Nov 2009.
Acknowledgments
The authors thank Jennifer Coward, Independent Medical Writer, Bollington, UK, for the editorial assistance with the preparation of this manuscript and Werner Wosniok, Department of Biomedical Statistics, University of Bremen, Germany, for statistical counseling. Financial support for the study was provided by an unrestricted grant by GlaxoSmithKline Biologicals and Sanofi Pasteur MSD.
Conflict of interest
P. Kaiser received a grant for this study from GSK and Sanofi Pasteur, and speakers’ grants for lectures from both companies. M. Borte reports consultant fees from Pfizer, Novartis, GlaxoSmithKline, and Sanofi Pasteur MSD and support for scientific meetings from Pfizer, Abbott, AstraZenica, GlaxoSmithKline, and Sanofi Pasteur MSD. K.P. Zimmer is on the national advisory board on rotavirus vaccination for Sanofi Pasteur MSD. H.I. Huppertz has served as a consultant and obtained speaking fees from GSK and Sanofi Pasteur MSD.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 71 kb)
Rights and permissions
About this article
Cite this article
Kaiser, P., Borte, M., Zimmer, KP. et al. Complications in hospitalized children with acute gastroenteritis caused by rotavirus: a retrospective analysis. Eur J Pediatr 171, 337–345 (2012). https://doi.org/10.1007/s00431-011-1536-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-011-1536-0