Abstract
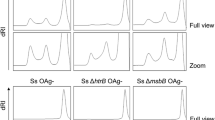
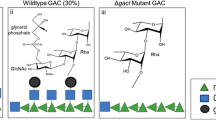
Inner core lipopolysaccharide (LPS) has been shown to be conserved in the majority of veterinary strains from the species Mannheimia haemolytica, Actinobacillus pleuropneumoniae and Pasteurella multocida and as such is being considered as a possible vaccine antigen. The proof-in-principle that a LPS-based antigen could be considered as a vaccine candidate has been demonstrated from studies with monoclonal antibodies raised to the inner core LPS of Mannheimia haemolytica, which were shown to be both bactericidal and protective in a mouse model of disease. In this study we confirm and extend the candidacy of the inner core LPS by demonstrating that it is possible to elicit functional antibodies against Mannheimia haemolytica wild-type strains following immunisation of rabbits with glycoconjugates elaborating the conserved inner core LPS antigen. The present study describes a conjugation strategy that uses amidases produced by Dictyostelium discoideum, targeting the amino functionality created by the amidase activity as the attachment point on the LPS molecule. To protect the amino functionality on the phosphoethanolamine (PEtn) residue of the inner core, we developed a novel blocking and unblocking strategy with t-butyl oxycarbonyl. A maleimide-thiol linker strategy with the thiol linker on the carboxyl residues of the carrier protein and the maleimide linker on the carbohydrate resulted in a high loading of carbohydrates per carrier protein. Immunisation derived antisera from rabbits recognised fully extended Mannheimia haemolytica LPS and whole cells from serotypes 1 and 2, despite a somewhat immunodominant response to the linkers also being observed. Moreover, bactericidal activity was demonstrated to a strain elaborating the immunising carbohydrate antigen and crucially to wild-type cells of serotypes 1 and 2, thus further supporting the consideration of inner core LPS as a potential vaccine antigen to combat disease caused by Mannheimia haemolytica.









Similar content being viewed by others
References
Davies, R.L., Donachie, W.: Intra-specific diversity and host specificity within Pasteurella haemolytica based on variation of capsular polysaccharide, lipopolysaccharide and outer-membrane proteins. Microbiol. 142, 1895–1907 (1996)
Whitely, L.O., Maheswaran, S.K., Weiss, D.J., Ames, T.R., Kannan, M.S.: Pasteurella haemolytica A1 and bovine respiratory disease: pathogenesis. J. Vet. Int. Med. 6, 11–22 (1992)
Biberstein, E.L.: Biotyping and serotyping of Pasteurella haemolytica. Methods Microbiol. 10, 252–269 (1978)
Angen, O., Mutters, R., Caugant, D.A., Olsen, J.E., Bisgaard, M.: Taxonomic relationships of the [Pasteurella] haemolytica complex as evaluated by DNA-DNA hybridizations and 16S rRNA sequencing with proposal of Mannheimia haemolytica gen. nov., comb. nov., Mannheimia granulomatis comb. nov., Mannheimia glucosida sp. nov., Mannheimia ruminalis sp. nov. and Mannheimia varigena sp. nov. Int. J. Syst. Bacteriol. 49, 67–86 (1999)
Ali, Q., Davies, R.L., Parton, R., Coote, J.G., Gibbs, H.A.: Lipopolysaccharide heterogeneity in Pasteurella haemolytica isolates from cattle and sheep. J. Gen. Microbiol. 138, 2185–2195 (1992)
Pandher, K., Confer, A.W., Murphy, G.L.: Genetic and immunologic analyses of PlpE, a lipoprotein important in complement-mediated killing of Pasteurella haemolytica serotype 1. Infect. Immun. 66, 5613–5619 (1998)
McKerral, L.J., Lo, R.Y.: Construction and characterisation of an acapsular mutant of Mannheimia haemolytica A1. Infect. Immun. 70, 2622–2629 (2002)
Shewen, P.E., Wilkie, B.N.: Cytotoxin of Pasteurella haemolytica acting on bovine leukocytes. Infect. Immun. 35, 91–94 (1982)
Confer, A.W., Simons, K.R.: Effects of Pasteurella haemolytica lipopolysaccharide on selected functions of bovine leukocytes. Am. J. Vet. Res. 47, 154–157 (1986)
Li, J., Clinkenbeard, K.D.: Lipopolysaccharide complexes with Pasteurella haemolytica leukotoxin. Infect. Immun. 67, 2920–2927 (1999)
Shewen, P.E., Wilkie, B.N.: Vaccination of calves with leukotoxic culture supernatant from Pasteurella haemolytica. Can. J. Vet. Res. 52, 30–36 (1988)
Ayalew, S., Blackwood, E.R., Confer, A.W.: Sequence diversity of the immunogenic outer membrane lipoprotein PlpE from Mannheimia haemolytica serotypes 1, 2 and 6. Vet. Microbiol. 114, 260–268 (2006)
St. Michael, F., Brisson, J.-R., Larocque, S., Monteiro, M., Li, J., Jacques, M., Perry, M.B., Cox, A.D.: Structural analysis of the lipopolysaccharide derived core oligosaccharides of Actinobacillus pleuropneumoniae serotypes 1, 2, 5a and the genome strain 5b. Carbohydr. Res. 339, 1973–1984 (2004)
St. Michael, F., Vinogradov, E.V., Li, J., Cox, A.D.: Structural analysis of the lipooligosaccharide from Pasteurella multocida genome strain Pm70 and identification of the putative lipopolysaccharide glycosyltransferases. Glycobiology 15, 323–333 (2005)
St. Michael, F., Li, J., Vinogradov, E.V., Larocque, S., Harper, M., Cox, A.D.: Structural analysis of the lipopolysaccharide from Pasteurella multocida strain VP161: Identification of both Kdo-P and Kdo-Kdo species in the lipopolysaccharide. Carbohydr. Res. 340, 59–68 (2005)
Stmichael, F., Harper, M., John, M., Stupak, J., Vinogradov, E., Parnas, H., Boyce, J.D., Adler, B., Cox, A.D.: Structural and genetic basis for the serological differentiation of Pasteurella multocida Heddleston serotypes 2 and 5. J. Bacteriol. 191, 6950–6959 (2009)
Brisson, J.-R., Crawford, E., Uhrin, D., Khieu, N.H., Perry, M.B., Severn, W.B., Richards, J.C.: The core oligosaccharide component from Mannheimia (Pasteurella) haemolytica serotype Al lipopolysaccharide contains L-glycero-D-manno and D-glycero-D-manno-heptoses. Analysis of the structure and confirmation by high resolution NMR spectroscopy. Can. J. Chem. 80, 949–963 (2002)
Lawrence, P.K., Kittichotirat, W., Bumgarner, R.E., McDermott, J.E., Herndon, D.R., Knowles, D.P., Srikumaran, S.: Genome sequences of Mannheimia haemolytica serotype A2: ovine and bovine isolates. J. Bacteriol. 192, 1167–1168 (2010)
St. Michael, F., Vinogradov, E., Cox, A.D.: Structural analyses of the core oligosaccharide from the lipopolysaccharide of bovine and ovine strains of Mannheimia haemolytica serotype 2. Carbohydr. Res. 346, 1333–1336 (2011)
Logan, S.M., Chen, W., Aubry, A., Gidney, M.A.J., Lacelle, S., St. Michael, F., Kuolee, R., Higgins, M., Neufeld, S., Cox, A.D.: Production of a D-glycero-D-manno-heptosyltransferase mutant of Mannheimia haemolytica displaying a veterinary pathogen specific conserved LPS structure and development and cross-reactivity of antibodies to this LPS structure. Vet. Microbiol. 116, 175–186 (2006)
Cox, A.D., Zou, W., Gidney, M.A.J., Lacelle, S., Plested, J.S., Makepeace, K., Wright, J.C., Coull, P., Moxon, E.R., Richards, J.C.: Candidacy of LPS-based glycoconjugates to prevent invasive meningococcal disease: developmental chemistry and investigation of immunological responses following immunization of mice and rabbits. Vaccine 23, 5045–5054 (2005)
Cox, A.D., St. Michael, F., Cairns, C.M., Lacelle, S., Filion, A., Neelamegan, D., Wenzel, C.Q., Richards, J.C.: Investigating the potential of conserved inner core oligosaccharide regions of Moraxella catarrhalis lipopolysaccharide as vaccine antigens: accessibility and functional activity of monoclonal antibodies and glycoconjugate derived sera. Glycoconjugate J. 28, 165–182 (2011)
Ren, D., Yu, S., Gao, S., Peng, D., Petralia, R.S., Muszynski, A., Carlson, R.W., Robbins, J.B., Tsai, C.-M., Lim, D.J., Gu, X.-X.: Mutant lipooligosaccharide-based glycoconjugate vaccine demonstrates a broad-spectrum effectiveness against Moraxella catarrhalis. Vaccine 29, 4210–4217 (2011)
Claviez, M., Pagh, K., Maruta, H., Baltes, W., Fisher, P., Gerisch, G.: Electron microscopic mapping of monoclonal antibodies on the tail region of Dictyostelium myosin. EMBO J. 1, 1017–1022 (1982)
Sawardeker, D.G., Sloneker, J.H., Jeanes, A.: Quantitative determination of monosaccharides as their alditol acetates by gas liquid chromatography. Anal. Chem. 37, 1602–1604 (1965)
Cox, A.D., Li, J., Brisson, J.-R., Moxon, E.R., Richards, J.C.: Structural analysis of the lipopolysaccharide from Neisseria meningitidis strain BZ157 galE: localisation of two phosphoethanolamine residues in the inner core oligosaccharide. Carbohydr. Res. 337, 1435–1444 (2002)
Laemmli, U.K.: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970)
Sutherland, A.D.: A rapid micro-method for the study of antibody-mediated killing of bacteria, with specific application to infection of sheep with Pasteurella haemolytica. Vet. Microbiol. 16, 263–271 (1988)
Cox, A.D., St. Michael, F., Neelamegan, D., Lacelle, S., Cairns, C.M., Richards, J.C.: Investigating the candidacy of LPS-based glycoconjugates to prevent invasive meningococcal disease: chemical strategies to prepare glycoconjugates with good carbohydrate loading. Glycoconjugate J. 27, 401–17 (2010)
Verez-Bencomo, V., Fernández-Santana, V., Hardy, E., Toledo, M.E., Rodríguez, M.C., Heynngnezz, L., et al.: A synthetic conjugate polysaccharide vaccine against Haemophilus influenzae type B. Science 305, 522–525 (2004)
Cox, A.D., St. Michael, F., Neelamegan, D., Lacelle, S., Cairns, C.M., Giuliani, M.M., et al.: Investigating the candidacy of LPS-based glycoconjugates to prevent invasive meningococcal disease: immunology of glycoconjugates with high carbohydrate loading. Glycoconjugate J. 27, 643–648 (2010)
Lipinski, T., Luu, T., Kitov, P.I., Szpacenko, A., Bundle, D.R.: A structurally diversified linker enhances the immune response to a small carbohydrate hapten. Glycoconjugate J. 28, 149–164 (2011)
Acknowledgements
We thank Perry Fleming (core Bacterial Culture Facility) for large scale biomass production and Jacek Stupak for mass spectrometry. We also thank Drs. Srikumaran Subramaniam and William Foreyt, Washington State University and Anthony Confer, Oklahoma State University, for providing us with M. haemolytica serotype A2 isolates from domestic sheep and cattle, respectively. We thank Annie Aubry and Dr. Susan Logan for helpful discussions. We are grateful to Novartis Vaccines for providing us with CRM197.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
St. Michael, F., Cairns, C., Filion, A.L. et al. Investigating the candidacy of lipopolysaccharide-based glycoconjugates as vaccines to combat Mannheimia haemolytica . Glycoconj J 28, 397–410 (2011). https://doi.org/10.1007/s10719-011-9339-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-011-9339-0