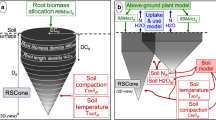
Abstract
Aims
Traits of the plant root system architecture (RSA) play a key role in crop performance. Therefore, architectural root traits are becoming increasingly important in plant phenotyping. In this study, we use a mathematical model to investigate the sensitivity of characteristic root system measures, obtained from different classical field root sampling schemes, to RSA parameters.
Methods
Root systems of wheat and maize were simulated and sampled virtually to mimic real field experiments using the root system architecture (RSA) model CRootBox. By means of a sensitivity analysis, we found RSA parameters that significantly influenced the virtual field sampling results. To identify correlations between sensitivities, we carried out a principal component analysis.
Results
We found that the parameters of zero order roots are the most sensitive, and parameters of higher order roots are less sensitive. Moreover, different characteristic root system measures showed different sensitivity to RSA parameters. RSA parameters that could be derived independently from different types of field observations were identified.
Conclusions
Selection of characteristic root system measures and parameters is essential to reduce the problem of parameter equifinality in inverse modeling with multi-parameter models and is an important step in the characterization of root traits from field observations.
















Similar content being viewed by others
Change history
15 August 2022
A Correction to this paper has been published: https://doi.org/10.1007/s11104-021-05089-3
References
Ayachit U (2015) The ParaView guide: a parallel visualization application, Kitware, ISBN 978-1930934306
Azevedo MCB, Chopart JL, Medina CC (2011) Sugarcane root length density and distribution from root intersection counting on a trench-profile. Sci Agric 68:94–101
Bardgett RD, Mommer L, De Vries FT (2014) Going underground: root traits as drivers of ecosystem processes. Trends Ecol Evol 29:692–699. https://doi.org/10.1016/j.tree.2014.10.006
Bingham IJ, Bengough AG (2003) Morphological plasticity of wheat and barley roots in response to spatial variation in soil strength. Plant Soil 250:273–282. https://doi.org/10.1023/A:1022891519039.
Bodner G, Nakhforoosh A, Kaul H-P (2015) Management of crop water under drought: a review. Agron Sustain Dev 35:401–442. https://doi.org/10.1007/s13593-015-0283-4
Bodner G, Alsalem M, Nakhforoosh A, Arnold T, Leitner D (2017) RGB and spectral root imaging for plant phenotyping and physiological research: experimental setup and imaging protocols. J Vis Exp:56251. https://doi.org/10.3791/56251
Bucksch A, Burridge J, York LM, Das A, Nord E, Weitz JS, Lynch JP (2014) Image-based high-throughput field phenotyping of crop roots. Plant Physiol 166:470–486
Cai G, Vanderborght J, Klotzsche A, van der Kruk J, Neumann J, Hermes N, Vereecken H (2016) Construction of Minirhizotron facilities for investigating root zone processes. Vadose Zone J 15. https://doi.org/10.2136/vzj2016.05.0043
Cai G, Vanderborght J, Langensiepen M, Schnepf A, Hüging H, Vereecken H (2018) Root growth, water uptake, and sap flow of winter wheat in response to different soil water conditions. Hydrol Earth Syst Sci 22:2449–2470. https://doi.org/10.5194/hess-22-2449-2018
Campolongo F, Cariboni J, Saltelli A (2007) An effective screening design for sensitivity analysis of large models. Environ Model Softw 22:1509–1518. https://doi.org/10.1016/j.envsoft.2006.10.004
Chen X, Ding Q, Błaszkiewicz Z, Sun J, Sun Q, He R, Li Y (2017) Phenotyping for the dynamics of field wheat root system architecture. Sci Rep 7:37649. https://doi.org/10.1038/srep37649https://www.nature.com/articles/srep37649#supplementary-information. Accessed 22 July 2018
Clark RT, MacCurdy RB, Jung JK, Shaff JE, McCouch SR, Aneshansley DJ, Kochian LV (2011) Three-dimensional root phenotyping with a novel imaging and software platform. Plant Physiol 156:455–465
Colombi T, Kirchgessner N, Walter A, Keller T (2017) Root tip shape governs root elongation rate under increased soil strength. Plant Physiol 174:2289–2301
Comas LH, Becker SR, Cruz VMV, Byrne PF, Dierig DA (2013) Root traits contributing to plant productivity under drought. Front Plant Sci 4:442. https://doi.org/10.3389/fpls.2013.00442
Couvreur V, Vanderborght J, Beff L, Javaux M (2014) Horizontal soil water potential heterogeneity: simplifying approaches for crop water dynamics models. Hydrol Earth Syst Sci 18:1723–1743. https://doi.org/10.5194/hess-18-1723-2014
de Dorlodot S, Forster B, Pagès L, Price A, Tuberosa R, Draye X (2007) Root system architecture: opportunities and constraints for genetic improvement of crops. Trends Plant Sci 12:474–481. https://doi.org/10.1016/j.tplants.2007.08.012
Dunbabin VM, Postma JA, Schnepf A, Pagès L, Javaux M, Wu L, Leitner D, Chen YL, Rengel Z, Diggle AJ (2013) Modelling root–soil interactions using three–dimensional models of root growth, architecture and function. Plant Soil 372:93–124. https://doi.org/10.1007/s11104-013-1769-y
Fan J, McConkey B, Wang H, Janzen H (2016) Root distribution by depth for temperate agricultural crops. Field Crop Res 189:68–74. https://doi.org/10.1016/j.fcr.2016.02.013
Fan Y, Miguez-Macho G, Jobbágy EG, Jackson RB, Otero-Casal C (2017) Hydrologic regulation of plant rooting depth. Proc Natl Acad Sci 114:10572–10577. https://doi.org/10.1073/pnas.1712381114
Feldman L (1994) The maize root. In: Freeling M, Walbot V (eds) The maize handbook. Springer New York, New York
Garré S, Pagès L, Laloy E, Javaux M, Vanderborght J, Vereecken H (2012) Parameterizing a dynamic architectural model of the root system of spring barley from Minirhizotron data. Vadose Zone J 11
Grabarnik P, Pagès L, Bengough AG (1998) Geometrical properties of simulated maize root systems: consequences for length density and intersection density. Plant Soil 200:157–167. https://doi.org/10.1023/A:1004382531671.
Gregory PJ (2006) Roots and the architecture of root systems. Plant roots. Blackwell Publishing Ltd, Oxford
Gregory PJ (2009) Measuring root system architecture: opportunities and challenges. In: International Symposium “Root Research and Applications” RootRAP, Vienna, Austria
Hamada A, Nitta M, Nasuda S, Kato K, Fujita M, Matsunaka H, Okumoto Y (2012) Novel QTLs for growth angle of seminal roots in wheat (Triticum aestivum L.). Plant Soil 354:395–405. https://doi.org/10.1007/s11104-011-1075-5
Hang LTT (2011) Quantifying root length density from soil auger samples: comparison of methods and effects of sample size. Institute of Organic Agriculture, Faculty of Agriculture. Rheinischen Friedrich-Wilhelms-Universität Bonn, Bonn
Hoad SP, Russell G, Kettlewell PS, Belshaw M (2004) Root system management in winter wheat: practices to increase water and nitrogen use. Agriculture and Horticulture Development Board, Kenilworth, Warwickshire
Hochholdinger F, Tuberosa R (2009) Genetic and genomic dissection of maize root development and architecture. Curr Opin Plant Biol 12:172–177. https://doi.org/10.1016/j.pbi.2008.12.002
Kalogiros DI, Adu MO, White PJ, Broadley MR, Draye X, Ptashnyk M, Bengough AG, Dupuy LX (2016) Analysis of root growth from a phenotyping data set using a density-based model. J Exp Bot 67:1045–1058. https://doi.org/10.1093/jxb/erv573
Kolb LN, Gallandt ER, Mallory EB (2017) Impact of spring wheat planting density, row spacing, and mechanical weed control on yield, grain protein, and economic return in Maine. Weed Sci 60:244–253. https://doi.org/10.1614/WS-D-11-00118.1
Kuijken RCP, van Eeuwijk FA, Marcelis LFM, Bouwmeester HJ (2015) Root phenotyping: from component trait in the lab to breeding. J Exp Bot 66:5389–5401. https://doi.org/10.1093/jxb/erv239.
Kutschera L (1960) Wurzelatlas mitteleuropäischer Ackerunkräuter und Kulturpflanzen. DLG Verlag, Frankfurt am Main
Landl M, Schnepf A, Vanderborght J, Bengough AG, Bauke SL, Lobet G, Bol R, Vereecken H (2018) Measuring root system traits of wheat in 2D images to parameterize 3D root architecture models. Plant Soil 425:457–477. https://doi.org/10.1007/s11104-018-3595-8
Leitner D, Klepsch S, Bodner G, Schnepf A (2010) A dynamic root system growth model based on L-systems. Plant Soil 332:177–192. https://doi.org/10.1007/s11104-010-0284-7
Leitner D, Felderer B, Vontobel P, Schnepf A (2014) Recovering root system traits using image analysis exemplified by two-dimensional neutron radiography images of lupine. Plant Physiol 164:24–35. https://doi.org/10.1104/pp.113.227892
Lipiec J, Siczek A, Sochan A, Bieganowski A (2016) Effect of sand grain shape on root and shoot growth of wheat seedlings. Geoderma 265:1–5. https://doi.org/10.1016/j.geoderma.2015.10.022
Lobet G, Pound MP, Diener J, Pradal C, Draye X, Godin C, Javaux M, Leitner D, Meunier F, Nacry P, Pridmore TP, Schnepf A (2015) Root system markup language: toward a unified root architecture description language. Plant Physiol 167:617–627. https://doi.org/10.1104/pp.114.253625
Lynch J (1995) Root architecture and plant productivity. Plant Physiol 109:7–13
Meunier F, Couvreur V, Draye X, Vanderborght J, Javaux M (2017) Towards quantitative root hydraulic phenotyping: novel mathematical functions to calculate plant-scale hydraulic parameters from root system functional and structural traits. J Math Biol 75:1133–1170. https://doi.org/10.1007/s00285-017-1111-z
Morris MD (1991) Factorial sampling plans for preliminary computational experiments. Technometrics 33:161–174. https://doi.org/10.2307/1269043
Muñoz-Romero V, Benítez-Vega J, López-Bellido RJ, Fontán JM, López-Bellido L (2010) Effect of tillage system on the root growth of spring wheat. Plant Soil 326:97–107. https://doi.org/10.1007/s11104-009-9983-3
Nagel KA, Kastenholz B, Jahnke S, van Dusschoten D, Aach T, Mühlich M, Truhn D, Scharr H, Terjung S, Walter A, Schurr U (2009) Temperature responses of roots: impact on growth, root system architecture and implications for phenotyping. Funct Plant Biol 36:947–959
Pagès L, Picon-Cochard C (2014) Modelling the root system architecture of Poaceae. Can we simulate integrated traits from morphological parameters of growth and branching? New Phytol 204:149–158. https://doi.org/10.1111/nph.12904
Pagès L, Vercambre G, Drouet J-L, Lecompte F, Collet C, Le Bot J (2004) Root Typ: a generic model to depict and analyse the root system architecture. Plant Soil 258:103–119. https://doi.org/10.1023/B:PLSO.0000016540.47134.03
Pagès L, Bruchou C, Garré S (2012) Links between root length density profiles and models of the root system architecture. Vadose Zone J 11
Pagès L, Xie J, Serra V (2013) Potential and actual root growth variations in root systems: modeling them with a two-step stochastic approach. Plant Soil 373:723–735. https://doi.org/10.1007/s11104-013-1820-z
Passot S, Couvreur V, Meunier F, Draye X, Javaux M, Leitner D, Pagès L, Schnepf A, Vanderborght J, Lobet G (2018) Connecting the dots between computational tools to analyse soil–root water relations. Journal of Experimental Botany:ery361–ery361. https://doi.org/10.1093/jxb/ery361
Perkons U, Kautz T, Uteau D, Peth S, Geier V, Thomas K, Lütke Holz K, Athmann M, Pude R, Köpke U (2014) Root-length densities of various annual crops following crops with contrasting root systems. Soil Tillage Res 137:50–57. https://doi.org/10.1016/j.still.2013.11.005
Ruano MV, Ribes J, Seco A, Ferrer J (2012) An improved sampling strategy based on trajectory design for application of the Morris method to systems with many input factors. Environ Model Softw 37:103–109. https://doi.org/10.1016/j.envsoft.2012.03.008
Schnepf A, Leitner D, Lobet G, Mai TH, Zörner M, Landl M, Sheng C, Morandage S, Vanderborght J, Vereecken H (2017) Triticum aestivum - CRootBox parameter file.figshare. Fileset. https://figshare.com/articles/Triticum_aestivum_-_CRootBox_parameter_file/4868438. Accessed 26 Aug 2017
Schnepf A, H K, L M, M F, Petrich L, Schmidt V (2018a) Statistical characterization of the root system architecture model CRootBox. Vadose Zone J 17. https://doi.org/10.2136/vzj2017.12.0212
Schnepf A, Leitner D, Landl M, Lobet G, Mai TH, Morandage S, Sheng C, Zörner M, Vanderborght J, Vereecken H (2018b) CRootBox: a structural–functional modelling framework for root systems. Annals of Botany:mcx221-mcx221. https://doi.org/10.1093/aob/mcx221
Schroth G, Kolbe D (1994) A method of processing soil core samples for root studies by subsampling. Biol Fertil Soils 18:60–62. https://doi.org/10.1007/BF00336446
Smit AL, George E, Groenwold J (2000) Root observations and measurements at (transparent) interfaces with soil. In: Smit AL, Bengough AG, Engels C, van Noordwijk M, Pellerin S, van de Geijn SC (eds) Root methods: a handbook. Heidelberg, Berlin, Heidelberg
Vansteenkiste J, Van Loon J, Garré S, Pagès L, Schrevens E, Diels J (2014) Estimating the parameters of a 3-D root distribution function from root observations with the trench profile method: case study with simulated and field-observed root data. Plant Soil 375:75–88. https://doi.org/10.1007/s11104-013-1942-3
Wasson AP, Richards RA, Chatrath R, Misra SC, Prasad SVS, Rebetzke GJ, Kirkegaard JA, Christopher J, Watt M (2012) Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J Exp Bot 63:3485–3498. https://doi.org/10.1093/jxb/ers111
Wasson AP, Rebetzke GJ, Kirkegaard JA, Christopher J, Richards RA, Watt M (2014) Soil coring at multiple field environments can directly quantify variation in deep root traits to select wheat genotypes for breeding. J Exp Bot 65:6231–6249. https://doi.org/10.1093/jxb/eru250
Weaver JE, Kramer J, Reed M (1924) Development of root and shoot of winter wheat under field environment. Ecology 5:26–50. https://doi.org/10.2307/1929162
Acknowledgements
This work was supported by the Transregional Collaborative Research Centre 32 (TR32), which is funded by the Deutsche Forschungsgemeinschaft (DFG)
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Anton Wasson.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. A. 1
Normalized means of absolute elementary effects (μ*) and standard deviations of elementary effects (σ) of the most sensitive RSA parameters on the maximum root length density in vertical root length density profiles derived from soil cores (RMc) first row), on the total root length per surface area derived from soil core measurements (Tc second row), and on the average root count density vertical profile walls (Tt third row). Blue line: 1:1 line, red line: μ* = 2 σ/sqrt(r) line (winter wheat (A) and maize (B) after 240 days and 180 days of simulation time respectively). (PNG 3917 kb)
Fig. A. 2
Normalized means of absolute elementary effects (μ*) and standard deviations of elementary effects (σ) of the RSA parameters on the depth above which 99% of the total root length is observed (D99) for winter wheat (left) and maize (right) 240 days and 180 days after sowing, respectively. Blue line: 1:1 line, red line: μ* = 2 σ/sqrt(r). (PNG 1403 kb)
Fig. A. 3
Normalized means of absolute elementary effects (μ*) of the most sensitive RSA parameters on root impact densities in vertical transects of trench profiles (RX) at different distances from the stem for winter wheat (left) and maize (right) 240 days and 180 days after sowing, respectively. (PNG 132 kb)
Fig. A. 4
Biplots of parameter loadings for different PCs when all data are combined for the wheat crop. (PNG 609 kb)
Fig. A. 5
Biplots of parameter loadings for different PCs when all data are combined for the maize crop. (PNG 4537 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morandage, S., Schnepf, A., Leitner, D. et al. Parameter sensitivity analysis of a root system architecture model based on virtual field sampling. Plant Soil 438, 101–126 (2019). https://doi.org/10.1007/s11104-019-03993-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-03993-3