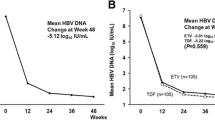
Abstract
Background
Entecavir, a drug with high potency and genetic barrier against hepatitis B virus, was believed to become very effective in reducing the hepatitis B burden in India. Long-term studies on its treatment outcome are scarce.
Methods
One hundred and six chronic hepatitis B (CHB) patients (17 cirrhotics of Child class A/B) who received entecavir therapy (0.5/1 mg/day) in a prospective open-label study from January 2010 to September 2015 were included in the analysis. Strict drug compliance was ensured. At least 1-year follow up was mandatory. Patients were followed up for HBV DNA negativity, e antigen seroconversion and hepatic events. Subgroup analysis for HBV DNA negativity was done for age (below and above 60 years), sex, HBV DNA level, e antigen status, cirrhosis and prior other modes of therapy.
Results
One (0.94 %) patient had primary drug resistance. Mean follow up was 2.5 (1 to 5) years. Overall HBV DNA negativity was 89 % to 98 % at 1 to 5 years and e antigen seroconversion rate 18.2 % at 5 years. ALT normalization paralleled HBV DNA negativity. No flare, decompensation, hepatocellular cancer or adverse reaction to drug was observed. Most achieved HBV DNA negativity after 6 months of therapy with lower response in those with high HBV DNA level, cirrhosis and prior therapy at baseline but only up to 1 year. Relapse was universal after stoppage of therapy. None lost HBsAg.
Conclusion
Entecavir will need to be continued indefinitely in Indian patients with CHB.
Similar content being viewed by others
References
World Health Organization. Hepatitis B. World Health Organization Fact Sheet No 204 (Updated July 2014) [accessed 2015 Dec 01]. Available at: http://www.who.int/mediacentre/factsheets/fs204/en/.
Chowdhury A, Santra A, Chakravorty A, et al. Community-based epidemiology of hepatitis B virus infection in West Bengal, India: prevalence of hepatitis B e antigen-negative infection and associated viral variants. J Gastroenterol Hepatol. 2005;20:1712–20.
Lian J, Han T, Xiang HL, et al. A randomized controlled trial on 240-week monotherapy with entecavir or adefovir in patients with chronic hepatitis B and cirrhosis. Zhonghua Gan Zang Bing Za Zhi. 2015;23:733–7.
Tseng KC, Tseng CW, Hsieh TY, et al. Efficacy of entecavir therapy for hepatitis B e-antigen positive chronic hepatitis B patients with prior exposure to interferon or nucleoside/nucleotide analogues. Hepatol Res. 2015 Sep 30. doi: 10.1111/hepr.12600. (Epub ahead of print)
Fong TL, Tien A, Jo KJ, et al. Durability of hepatitis B e antigen seroconversion in chronic hepatitis B patients treated with entecavir or tenofovir. Dig Dis Sci. 2015;60:3465–72.
Chang TT, Lai CL, Kew Yoon S, et al. Entecavir treatment for up to 5 years in patients with hepatitis B e antigen-positive chronic hepatitis B. Hepatology. 2010;51:422–30.
Lee HW, Park JY, Ahn SH. An evaluation of entecavir for the treatment of chronic hepatitis B infection in adults. Expert Rev Gastroenterol Hepatol. 2016;10:177–86.
Patra UC. An open label prospective observational study to evaluate the efficacy of entecavir in HBeAg positive treatment naïve chronic hepatitis B patients in a tertiary care center in eastern India. Trop Gastroenterol. 2011;32:314–6.
Goyal SK, Dixit VK, Shukla SK, et al. Prolonged use of tenofovir and entecavir in hepatitis B virus-related cirrhosis. Indian J Gastroenterol. 2015;34:286–91.
Ismail AM, Sharma OP, Kumar MS, et al. Virological response and antiviral resistance mutations in chronic hepatitis B subjects experiencing entecavir therapy: an Indian subcontinent perspective. Antiviral Res. 2013;98:209–16.
Choi HN, Song JE, Lee HC, et al. Efficacy of prolonged entecavir monotherapy in treatment-naïve chronic hepatitis B patients exhibiting a partial virologic response to entecavir. Clin Mol Hepatol. 2015;21:24–31.
Biçer KÇ, Ari A, Genç VE, et al. Efficacy of entecavir treatment among chronic hepatitis B nucleos(t)ide-naïve and -experienced patients. Turk J Med Sci. 2015;45:99–104.
Jo YJ, Kim KA, Lee JS, et al. Long-term virological outcome in chronic hepatitis B patients with a partial virological response to entecavir. Korean J Intern Med. 2015;30:170–6.
Wang JL, Du XF, Chen SL, et al. Histological outcome for chronic hepatitis B patients treated with entecavir vs lamivudine-based therapy. World J Gastroenterol. 2015;21:9598–606.
Park JH, Ahn SJ, Cho HJ, et al. Clinical course of partial virological responders under prolonged entecavir monotherapy in patients with chronic hepatitis B. J Med Virol. 2016;88:252–9.
Lee HW, Kwon JC, Oh IS, et al. Prolonged entecavir therapy is not effective for HBeAg seroconversion in treatment-naive chronic hepatitis B patients with a partial virological response. Antimicrob Agents Chemother. 2015;59:5348–56.
Yan LB, Chen EQ, Bai L, et al. Efficacy of entecavir treatment for up to 96 weeks in nucleoside-naive HBeAg-positive chronic hepatitis B patients with high viral load. Clin Res Hepatol Gastroenterol. 2015;39:366–72.
Idilman R, Gunsar F, Koruk M, et al. Long-term entecavir or tenofovir disoproxil fumarate therapy in treatment-naïve chronic hepatitis B patients in the real-world setting. J Viral Hepat. 2015;22:504–10.
Sohn HR, Min BY, Song JC, et al. Off-treatment virologic relapse and outcomes of re-treatment in chronic hepatitis B patients who achieved complete viral suppression with oral nucleos(t)ide analogs. BMC Infect Dis. 2014;14:439.
Tan YW, Ge GH, Sun L, et al. Efficacy of Entecavir therapy in elderly patients with chronic hepatitis B infection. Braz J Infect Dis. 2014;18:691–2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
GR declares that he has no conflict of interest.
Ethics statement
All procedures performed in the study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Ray, G. 5-year efficacy of entecavir in Indian patients with chronic hepatitis B. Indian J Gastroenterol 35, 190–194 (2016). https://doi.org/10.1007/s12664-016-0664-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-016-0664-x