Abstract
Metal oxide nanotubes are believed to be promising materials with adsorption functionality for water purification due to their synergistic effect of the overall microscale morphology for easy separation and nanoscale surface characters providing enough surface active absorption sites. This work shows the synthesis of uniform hierarchical porous CeO2 nanotubes via nanowire-directed templating method and describes the adsorption behavior of CeO2 nanotubes for a typical azo dye Congo red which has resistance to oxidation and decoloration in natural conditions. Fourier transform infrared spectroscopy spectra provided the evidence that Congo red was successfully coated on the surface of CeO2 nanotubes by both bidentate-type bridge link of Ce4+ cations from sulfonate SO3 − groups and the electrostatic attraction between the protonated surface generated by oxygen vacancies and dissociated sulfonate groups. The adsorption kinetic data fitted well to the pseudo-second-order kinetic equation, whereas the Langmuir isotherm equation exhibited better correlation with the experimental data. The calculated maximum adsorption capacity from the isothermal model was 362.32 mg/g. In addition, the prepared CeO2 nanotubes exhibited good recyclability and reusability as highly efficient adsorbents for Congo red removal after regeneration. These favorable performances enable the obtained CeO2 nanotubes to be promising materials for dye removal from aqueous solution.
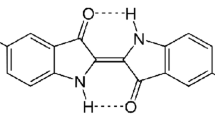
Graphical Abstract
CeO2 nanotubes composed of crystallized nanoparticles exhibit well adsorption ability for a typical azo dye Congo red.











Similar content being viewed by others
References
Abi-aad E, Bechara R, Grimblot J, Aboukais A (1993) Preparation and characterization of ceria under an oxidizing atmosphere. Thermal analysis, XPS, and EPR study. Chem Mater 5:793–797
Adebayo MA, Prola LDT, Lima EC, Puchana-Rosero MJ, Cataluna R, Saucier C, Umpierres CS, Vaghetti JCP, Silva LG, Ruggiero R (2014) Adsorption of Procion Blue MX-R dye from aqueous solutions by lignin chemically modified with aluminium and manganese. J Hazard Mater 268:43–50
Azizian S (2004) Kinetic models of sorption: a theoretical analysis. J Colloid Interface Sci 276:47–52
Bai YY, Lu Y, Liu JK (2016) An efficient photocatalyst for degradation of various organic dyes: Ag@Ag2MoO4–AgBr composite. J Hazard Mater 307:26–35
Calagui MJC, Senoro DB, Kan CC, Salvacion JWL, Futalan CM, Wane MW (2014) Adsorption of indium(III) ions from aqueous solution using chitosan-coated bentonite beads. J Hazard Mater 277:120–126
Castaldi P, Silvetti M, Garau G, Demurtas D, Deiana S (2015) Copper(II) and lead(II) removal from aqueous solution by water treatment residues. J Hazard Mater 283:140–147
Chen G, Sun S, Sun X, Fan W, You T (2009) Formation of CeO2 Nanotubes from Ce(OH)CO3 nanorods through Kirkendall diffusion. Inorg Chem 48:1334–1338
Chen S, Zhang J, Zhang C, Yue Q, Li Y, Li C (2010) Equilibrium and kinetic studies of methyl orange and methyl violet adsorption on activated carbon derived from Phragmites australis. Desalination 252:149–156
Chen C, Li P, Wang G, Yu Y, Duan F, Chen C, Song W, Qin Y, Knez M (2013) Nanoporous nitrogen-doped titanium dioxide with excellent photocatalytic activity under visible light irradiation produced by molecular layer deposition. Angew Chem Int Ed 52:9196–9200
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 97:1061–1085
Dias FF, Oliveira AAS, Arcanjo AP, Moura FCC, Pacheco JGA (2016) Residue-based iron catalyst for the degradation of textile dye via heterogeneous photo-Fenton. Appl Catal B Environ 186:136–142
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156:2–10
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Ghosh S, Kouame NA, Ramos L, Remita S, Dazzi A, Deniset-Besseau A, Beaunier P, Goubard F, Aubert PH, Remita H (2015) Conducting polymer nanostructures for photocatalysis under visible light. Nat Mater 14:505–511
Gong J, Liu J, Chen XC, Jiang ZW, Wen W, Mijowska E, Tang T (2015) Converting real-world mixed waste plastics into porous carbon nanosheets with excellent performance in the adsorption of an organic dye from wastewater. J Mater Chem A 3:341–351
Gupta VK, Suhas (2009) Application of low-cost adsorbents for dye removal—a review. J Environ Manage 90:2313–2342
Han WQ, Wu L, Zhu Y (2005) Formation and oxidation state of CeO2−x nanotubes. J Am Chem Soc 127:12814–12815
Han X, Tian P, Pang H, Song Q, Ning G, Yu Y, Fang H (2014) Facile synthesis of magnetic hierarchical MgO–MgFe2O4 composites and their adsorption performance towards Congo red. RSC Adv 4:28119–28125
Hernandez-Ramirez O, Holmes SM (2008) Novel and modified materials for wastewater treatment applications. J Mater Chem 18:2751–2761
Hu JS, Zhong LS, Song WG, Wan LJ (2008) Synthesis of hierarchically structured metal oxides and their application in heavy metal ion removal. Adv Mater 20:2977–2982
Hu J, Yu H, Dai W, Yan X, Hu X, Huang H (2014) Enhanced adsorptive removal of hazardous anionic dye “congo red” by a Ni/Cu mixed-component metal–organic porous material. RSC Adv 4:35124–35130
Ji P, Zhang J, Chen F, Anpo M (2009) Study of adsorption and degradation of acid orange 7 on the surface of CeO2 under visible light irradiation. Appl Catal B Environ 85:148–154
Juang RS, Wu FC, Tseng RL (1996) Adsorption isotherms of phenolic compounds from aqueous solutions onto activated carbon fibers. J Chem Eng Data 41:487–492
Kannan N, Sundaram MM (2001) Kinetics and mechanism of removal of methylene blue by adsorption on various carbons-a comparative study. Dyes Pigm 51:25–40
Karan CK, Bhattacharjee M (2016) Self-healing and moldable metallogels as the recyclable materials for selective dye adsorption and separation. ACS Appl Mater Interfaces 8:5526–5535
Kumar R, Rashid J, Barakat MA (2014) Synthesis and characterization of a starch–AlOOH–FeS2 nanocomposite for the adsorption of congo red dye from aqueous solution. RSC Adv 4:38334–38340
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Li ZC, Yang BJ, Zhang SN, Wang BN, Xue B (2014) A novel approach to hierarchical sphere-like ZnAl-layered double hydroxides and their enhanced adsorption capability. J Mater Chem A 2:10202–10210
Li DW, Chen DZ, Yao YY, Lin JC, Gong F, Wang L, Luo LS, Huang ZF, Zhang L (2016) Strong enhancement of dye removal through addition of sulfite to persulfate activated by a supported ferric citrate catalyst. Chem Eng J 288:806–812
Lian G, Zhang X, Si HB, Wang J, Cui DL, Wang QL (2013) Boron nitride ultrathin fibrous nanonets: one-step synthesis and applications for ultrafast adsorption for water treatment and selective filtration of nanoparticles. ACS Appl Mater Interfaces 5:12773–12778
Liang HW, Cao X, Zhang WJ, Lin HT, Zhou F, Chen LF, Yu SH (2011) Robust and highly efficient free-standing carbonaceous nanofiber membranes for water purification. Adv Funct Mater 21:3851–3858
Liu W, Ni JR, Yin XC (2014) Synergy of photocatalysis and adsorption for simultaneous removal of Cr(VI) and Cr(III) with TiO2 and titanate nanotubes. Water Res 53:12–25
Lu X, Zheng D, Gan J, Liu Z, Liang C, Liu P, Tong Y (2010) Porous CeO2 nanowires/nanowire arrays: electrochemical synthesis and application in water treatment. J Mater Chem 20:7118–7122
Maksimchuk PO, Masalov AA, Malyukin YV (2013) Spectroscopically detected formation of oxygen vacancies in nano-crystalline CeO2−x. J Nano Electron Phys 5:01004
Ouyang X, Li W, Xie S, Zhai T, Yu M, Gan J, Lu X (2013) Hierarchical CeO2 nanospheres as highly-efficient adsorbents for dye removal. New J Chem 37:585–588
Pan B, Xing BS (2015) Adsorption mechanisms of organic chemicals on carbon nanotubes. Environ Sci Technol 42:9005–9013
Pfau A, Schierbaum KD (1994) The electronic structure of stoichiometric and reduced CeO2 surfaces: an XPS, UPS and HREELS study. Surf Sci 321:71–80
Raven KP, Jain A, Loeppert RH (1998) Arsenite and arsenate adsorption on ferrihydrite: kinetics, equilibrium, and adsorption envelopes. Environ Sci Technol 32:344–349
Salleh MAM, Mahmoud DK, Karim WA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280:1–13
Santamaria M, Asaro L, Bocchetta P, Megna B, Di Quarto F (2013) Electrodeposition of CeO2 and Co-Doped CeO2 nanotubes by cyclic anodization in porous alumina membranes electrochemical/electroless deposition. ECS Electrochem Lett 2:D29–D32
Shalom M, Inal S, Fettkenhauer C, Neher D, Antonietti M (2013) Improving carbon nitride photocatalysis by supramolecular preorganization of monomers. J Am Chem Soc 135:7118–7121
Shen Y, Fang Q, Chen B (2015) Environmental applications of three-dimensional graphene-based macrostructures: adsorption, transformation, and detection. Environ Sci Technol 49:67–84
Shiue A, Ma CM, Ruan RT, Chang CT (2012) Adsorption kinetics and isotherms for the removal methyl orange from wastewaters using copper oxide catalyst prepared by the waste printed circuit boards. Sustain Environ Res 22:209–215
Si R, Zhang YW, Li SJ, Lin BX, Yan CH (2004) Urea-based hydrothermally derived homogeneous nanostructured Ce1−xZrxO2 (x = 0–0.8) solid solutions: a strong correlation between oxygen storage capacity and lattice strain. J Phys Chem B 108:12481–12488
Srivastav AL, Singh PK, Srivastava V, Sharma YC (2013) Application of a new adsorbent for fluoride removal from aqueous solutions. J Hazard Mater 263:342–352
Sun C, Li H, Chen L (2012) Nanostructured ceria-based materials: synthesis, properties, and applications. Energy Environ Sci 5:8475–8505
Tesh SJ, Scott TB (2014) Nano-composites for water remediation: a review. Adv Mater 26:6056–6068
Tomic NM, Dohcevic-Mitrovic ZD, Paunovic NM, Mijin DZ, Radic ND, Grbic BV, Askrabic SM, Babic BM, Bajuk-Bogdanovic DV (2014) Nanocrystalline CeO2−δ as effective adsorbent of azo dyes. Langmuir 30:11582–11590
Tseng RL, Wu FC (2008) Inferring the favorable adsorption level and the concurrent multi-stage process with the Freundlich constant. J Hazard Mater 155:277–287
Vinodgopal K, Bedja I, Kamat PV (1996) Nanostructured semiconductor films for photocatalysis. photoelectrochemical behavior of SnO2/TiO2 composite systems and its role in photocatalytic degradation of a textile azo dye. Chem Mater 8:2180–2187
Wang S, Zhang J, Jiang J, Liu R, Zhu B, Xu M, Wang Y, Cao J, Li M, Yuan Z, Zhang S, Huang W, Wu S (2009) Porous ceria hollow microspheres: synthesis and characterization. Microporous Mesoporous Mater 123:349–353
Wang T, Zhang L, Zhang J, Hua G (2013) Synthesis and characterization of mesoporous CeO2 nanotube arrays. Microporous Mesoporous Mater 171:196–200
Wang MX, Zhang QL, Yao SJ (2015) A novel biosorbent formed of marine-derived penicillium janthinellum mycelial pellets for removing dyes from dye-containing wastewater. Chem Eng J 259:837–844
Wang XS, Liang J, Li L, Lin ZJ, Bag PP, Gao SY, Huang YB, Cao R (2016) An anion metal-organic framework with lewis basic sites-rich toward charge-exclusive cationic dyes separation and size-selective catalytic reaction. Inorg Chem 55:2641–2649
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div Am Soc Civ Eng 89(SA2):31–40
Wu JS, Wang JS, Li HY, Du YC, Huang KL, Liu BX (2013a) Designed synthesis of hematite-based nanosorbents for dye removal. J Mater Chem A 1:9837–9847
Wu JS, Li HY, Wang JS, Li ZF (2013b) Swift adsorptive removal of congo red from aqueous solution by K1.33Mn8O16 nanowires. J Nanosci Nanotechnol 13:5452–5460
Wu ZJ, Xiong Y, Guan GQ, Kong LJ, Tian SH (2014) Preparation and adsorption behavior of new hollow-like spherical sludge chars for methylene blue. RSC Adv 4:55256–55262
Xu P, Zeng GM, Huang DL, Feng CL, Hu S, Zhao MH, Lai C, Wei Z, Huang C, Xie GX, Liu ZF (2012) Use of iron oxide nanomaterials in wastewater treatment: a review. Sci Total Environ 424:1–10
Yan B, Zhu H (2008) Controlled synthesis of CeO2 nanoparticles using novel amphiphilic cerium complex precursors. J Nanopart Res 10:1279–1285
Yang Z, Wang Q, Wei S, Ma D, Sun Q (2010) The effect of environment on the reaction of water on the ceria(111) surface: a DFT + U study. J Phys Chem C 114:14891–14899
Yang HY, Han ZJ, Yu SF, Pey KL, Ostrikov K, Karnik R (2013) Carbon nanotube membranes with ultrahigh specific adsorption capacity for water desalination and purification. Nature Commun 4:2220
Yu SJ, Wang XX, Ai YJ, Tan XL, Hayat T, Hu WP, Wang XK (2016) Experimental and theoretical studies on competitive adsorption of aromatic compounds on reduced graphene oxides. J Mater Chem A 4:5654–5662
Zhai T, Xie S, Lu X, Xiang L, Yu M, Li W, Liang C, Mo C, Zeng F, Tong TLY (2012) Porous Pr(OH)3 nanostructures as high-efficiency adsorbents for dye removal. Langmuir 28:11078–11085
Zhao GX, Jiang L, Yudong He, Li JX, Dong HL, Wang XK (2011) Sulfonated graphene for persistent aromatic pollutant management. Adv Mater 23:3959–3963
Zhao DL, Sheng GD, Chen CL, Wang XK (2012) Enhanced photocatalytic degradation of methylene blue under visible irradiation on graphene@TiO2 dyade structure. Appl Catal B Environ 111–112:303–308
Zhou J, Yang S, Yu J, Shu Z (2011) Novel hollow microspheres of hierarchical zinc–aluminum layered double hydroxides and their enhanced adsorption capacity for phosphate in water. J Hazard Mater 192:1114–1121
Zhu YP, Liu YL, Ren TZ, Yuan ZY (2014) Hollow manganese phosphonate microspheres with hierarchical porosity for efficient adsorption and separation. Nanoscale 6:6627–6636
Acknowledgments
We thank National Natural Science Foundation of China (51225402, 51402008 and 51471006), Beijing Natural Science Foundation (2142008, 2154043), and Guangxi Natural Science Foundation (2014GXNSFBA118039, 2014GXNSFBB118001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, J., Wang, J., Du, Y. et al. Adsorption mechanism and kinetics of azo dye chemicals on oxide nanotubes: a case study using porous CeO2 nanotubes. J Nanopart Res 18, 191 (2016). https://doi.org/10.1007/s11051-016-3497-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-016-3497-8