Abstract
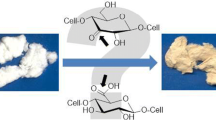
Studies of cellulose aging and yellowing that involved a 1,4-dimethyl 3-keto β-d-glucoside (compound 1) model of oxidized cellulose led to a hemi-hydrated crystal of title compound 2 (1,6-dihydroxy-3,8-bis-hydoxymethyl-5,10-dimethoxy-4,9-dioxa-tricyclo [5,3,1,1,2,6] dodecane-11,12-dione). The same compound 2 was isolated in low yield from “real-world” oxidized and aged cellulosic pulp. Formation of compound 2 implies cellulose chain cleavage and, unexpectedly, cross-linking during aging. X-ray diffraction revealed an encompassing 10-membered ring whose two carbonyl group bridges define two eight-membered rings and three six-membered rings. The central six-membered ring is antecedent to 2,5-dihydroxy-1,4-benzoquinone (compound 3), a potent and nearly ubiquitous chromophore in aged cellulose; the outer rings derive from the keto-glucosides and have 4C1 and 1C4 shapes with gt and rare tg O-6 positions. Weak trans-annular interactions between >C=O carbon and ring oxygen atoms were confirmed with Atoms-in-Molecules theory. That theory was also used to analyze a questionable cyclic hydrogen bond and bonds between adjacent O–H and carbonyl oxygens.





Similar content being viewed by others
References
Adorjan I, Rosenau T, Potthast A, Kosma P, Mereiter K, Pauli J, Jäger C (2004) Crystal and molecular structure of methyl 4-o-methyl-β-d-ribo-hex-3-ulopyranoside. Carbohydr Res 339:795–799
Adorjan I, Potthast A, Rosenau T, Sixta H, Kosma P (2005) Discoloration of cellulose solutions in N-methylmorpholine-N-oxide (Lyocell). Part 1: studies on model compounds and pulps. Cellulose 12:51–57
Ahn K, Henniges U, Banik G, Potthast A (2012) Is cellulose degradation due to β-elimination processes a threat in mass deacidification of library books? Cellulose 19:1149–1159
Bader RFW (1990) Atoms in molecules: a quantum theory. International series of monographs on chemistry. Oxford Clarendon Press, New York
Bürgi HB, Dunitz JD, Shefter E (1974) Chemical reaction paths. IV. Aspects of O···C=O interactions in crystals. Acta Cryst Sect B 30:1517–1527
Coppens P (2005) Charge densities come of age. Angew Chem Int Ed 44:6810–6811
Destro R, Merati F (1995) Bond lengths, and beyond. Acta Crystallogr Sect B 51:559–570
Fedeli W, Dunitz JD (1968) Die Strukturen der mittleren Ringverbindungen XV. 6-Ketononanolid (Oxacyclodeca-2,7-dion). Helv Chim Acta 51:445–458
French AD, Csonka GI (2011) Hydroxyl orientations in cellobiose and other polyhydroxyl compounds: modeling versus experiment. Cellulose 18:897–909
Groom CR, Allen FH (2014) The Cambridge structural database in retrospect and prospect. Angew Chem Int Ed 53:662–671
Hosoya T, French AD, Rosenau T (2013) Chemistry of 5,8-dihydroxy-[1,4]-naphthoquinone, a key chromophore in aged cellulosics. Mini Rev Org Chem 10:309–315
Hübschle CB, Luger P (2006) Moliso—a program for colour-mapped iso-surfaces. J Appl Cryst 39:901–904
Keith TA (2010) AIMAll, http://aim.tkgristmill.com/references.html
Korntner P, Hosoya T, Dietz T, Eibinger K, Reiter H, Spitzbart M, Röder T, Borgads A, Kreiner W, Mahler AK, Winter H, Groiss Y, Wong S, French AD, Henniges U, Potthast A, Rosenau T (2015) Chromophores in lignin-free cellulosic materials belong to three compound classes. Chromophores in cellulosics XII. Cellulose 22:1053–1062
Macrae F, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez-Monge L, Taylor R, van de Streek J, Wood PA (2008) Mercury CSD 2.0—new features for the visualization and investigation of crystal structures. J Appl Cryst 41:466–470
Nishiyama Y, Kuga S, Okano T (2000) Mechanism of mercerization revealed by X-ray diffraction. J Wood Sci 46:452–457
Olsson K, Pernemalm PA, Theander O (1978) Formation of aromatic compounds from carbohydrates. VII. Reaction of d-glucose and glycine in slightly acidic, aqueous solution. Acta Chem Scand B 32:249–256
Owicki JC, Shipman LL, Scheraga HA (1975) Structure, energetics, and dynamics of small water clusters. J Phys Chem 79:1794–1811
Popoff T, Theander O (1972) Formation of aromatic compounds from carbohydrates: part 1. Reaction of d-glucuronic acid, d-glacturonic acid, d-xylose, and l-arabinose in slightly acidic, aqueous solution. Carbohydr Res 22:135–149
Potthast A, Röhrling J, Rosenau T, Borgards A, Sixta H, Kosma P (2003) A novel method for the determination of carbonyl groups in cellulosics by fluorescence labeling. 3. Monitoring oxidative processes. Biomacromolecules 4:743–749
Potthast A, Rosenau T, Kosma P, Saariaho A-M, Vuorinen T (2005) On the nature of carbonyl groups in cellulosic pulps. Cellulose 12:43–50
Řezáč J, Riley KE, Hobza P (2011) S66: a well-balanced database of benchmark interaction energies relevant to biomolecular structures. J Chem Theory Comput 7:2427–2438
Röhrling J, Potthast A, Rosenau T, Lange T, Ebner G, Sixta H, Kosma P (2002) A novel method for the determination of carbonyl croups in cellulosics by fluorescence labeling. 1. Method development. Biomacromolecules 3:959–968
Rosenau T, Potthast A, Milacher W, Adorjan I, Hofinger A, Kosma P (2005) Discoloration of cellulose solutions in N-methylmorpholine-N-oxide (Lyocell). Part 2: isolation and identification of chromophores. Cellulose 12:197–208
Rosenau T, Potthast A, Krainz K, Yoneda Y, Dietz T, Peralta-Inga Shields Z, French AD (2011) Chromophores in cellulosics, VI. First isolation and identification of residual chromophores from aged cotton linters. Cellulose 18:1623–1633
Singh UC, Kollman PA (1985) A water dimer potential based on ab initio calculations using Morokuma component analyses. J Chem Phys 83:4033–4040
Xu X, Goddard III WA (2004) The X3LYP extended density functional for accurate descriptions of nonbond interactions, spin states, and thermochemical properties. Proc Natl Acad Sci USA 101:2673–2677
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rosenau, T., Potthast, A., Kosma, P. et al. 2,4′:2′,4 Dianhydride of 3-keto-glucoside, a precursor to chromophores of aged, yellow cellulose, and its weak interactions. Cellulose 24, 1227–1234 (2017). https://doi.org/10.1007/s10570-017-1198-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-017-1198-9